Unit 1: Subatomic Organization and Spectroscopic Evidence
Atomic Structure and Electron Configuration
Understanding how atoms are constructed is the foundation of chemistry. In AP Chemistry, we move beyond the simple Bohr model (orbits) to the Quantum Mechanical model (orbitals), using mathematical probability to determine where electrons are located and how they behave.
The Quantum Mechanical Model
The current model of the atom describes electrons not as particles circling the nucleus like planets, but as strictly defined energy states described by wave functions. The location of an electron is defined by orbitals—regions of space where there is a high probability (90%) of finding an electron.
Coulomb’s Law: The Governing Force
Before diving into orbitals, you must understand the force holding the atom together. Coulomb’s Law describes the electrostatic interaction between electrically charged particles.
Where:
- $Q1$ and $Q2$ are the magnitudes of the charges (nucleus (+) and electron (-)).
- $r$ is the distance between the centers of the charged particles.
Key Takeaways for AP Chem:
- Magnitude of Charge: A more positive nucleus (higher atomic number/higher Z) exerts a stronger pull on electrons (higher ionization energy), assuming distance ($r$) remains roughly the same.
- Distance: Electrons further from the nucleus ($r$ increases) experience a weaker attractive force and have higher potential energy (they are easier to remove).
Electron Configurations and Quantum Numbers
To describe the "address" of an electron, we use four quantum numbers, though AP Chemistry focuses primarily on the arrangement resulting from them.
- Principal Quantum Number ($n$): Indicates the main energy level or shell. Corresponds to the periods (rows) on the periodic table ($n=1, 2, 3…$). As $n$ increases, the average distance from the nucleus increases.
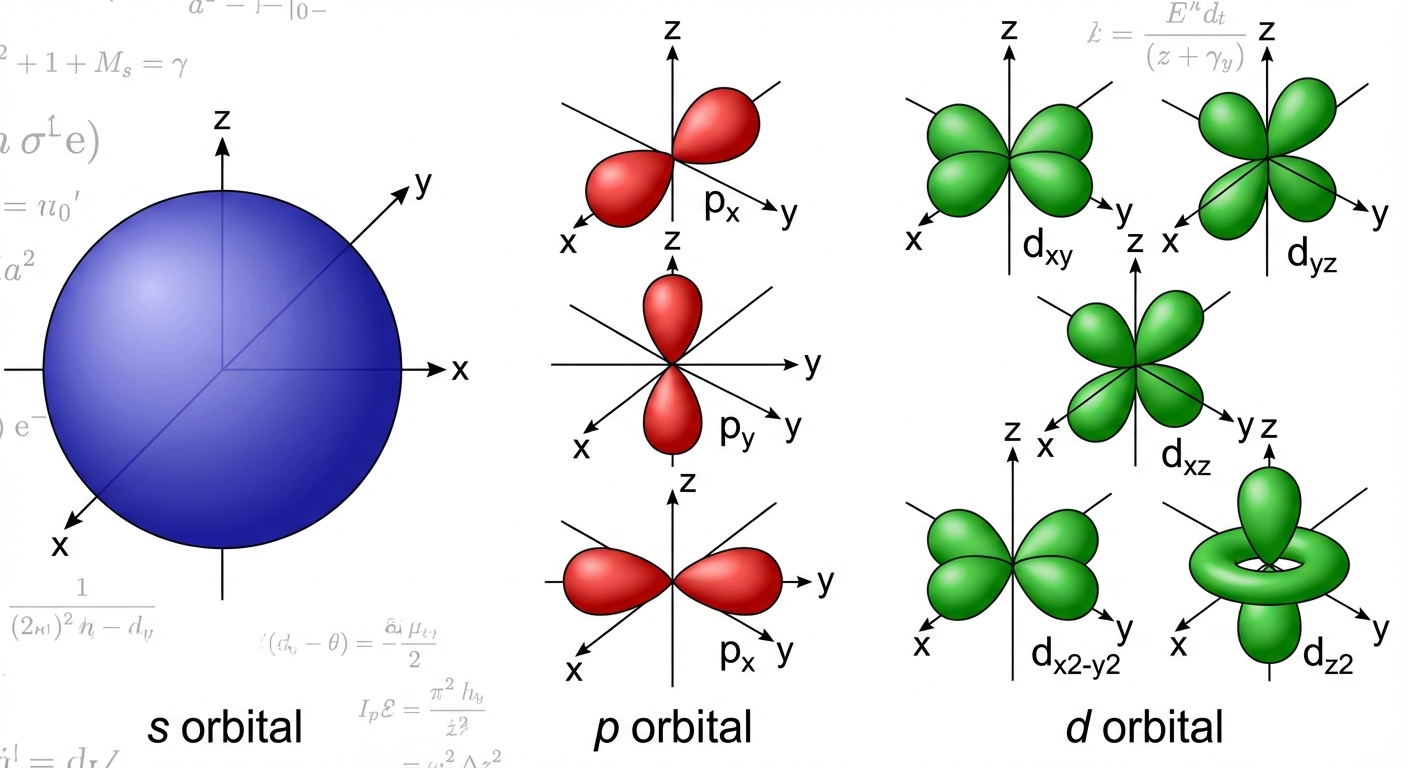
- Angular Momentum Quantum Number ($l$): Indicates the shape of the orbital (subshell).
| Subshell | Shape | Max Electrons | Starts at Level |
|---|---|---|---|
| s | Sphere | 2 | $n=1$ |
| p | Dumbbell (Peanut) | 6 | $n=2$ |
| d | Cloverleaf (Double Dumbbell) | 10 | $n=3$ |
| f | Complex | 14 | $n=4$ |
Rules for Filling Orbitals
When writing electron configurations, three rules govern how electrons populate the atom:
- The Aufbau Principle: Electrons fill the lowest energy orbitals first. (e.g., $1s$ fills before $2s$).
- Note: The $4s$ subshell is slightly lower in energy than the $3d$ subshell when empty, so $4s$ fills before $3d$.
- ** The Pauli Exclusion Principle:** No two electrons in an atom can have the same four quantum numbers. Practically, this means an orbital can hold a maximum of 2 electrons, and they must have opposite spins (represented as up $\uparrow$ and down $\downarrow$ arrows).
- Hund’s Rule: For degenerate orbitals (orbitals of the same energy, like the three $2p$ orbitals), electrons fill each orbital singly with parallel energy spins before pairing up.
Writing Configurations
There are two main ways to represent electron arrangements:
1. Full Electron Configuration:
List every subshell in order of increasing energy.
- Example (Oxygen, 8 e-): $1s^2 2s^2 2p^4$
- Example (Iron, 26 e-): $1s^2 2s^2 2p^6 3s^2 3p^6 4s^2 3d^6$
2. Noble Gas (Condensed) Notation:
Use the preceding noble gas to represent core electrons.
- Example (Iron): $[Ar] 4s^2 3d^6$
Anomalies and Exceptions
Nature prefers symmetry and stability. Half-filled and fully-filled $d$-subshells offer extra stability. You must memorize these two common exceptions:
- Chromium (Cr): Expected: $[Ar] 4s^2 3d^4$ $\rightarrow$ Actual: $[Ar] 4s^1 3d^5$ (Promotes one $s$ electron to make $d$ half-filled).
- Copper (Cu): Expected: $[Ar] 4s^2 3d^9$ $\rightarrow$ Actual: $[Ar] 4s^1 3d^{10}$ (Promotes one $s$ electron to make $d$ fully-filled).
Configuration of Ions
When forming cations (positive ions) from transition metals, electrons are removed from the valence shell with the highest $n$ value first.
Crucial Rule: Remove electrons from the s-orbital before the d-orbital.
- Fe Atom: $[Ar] 4s^2 3d^6$
- Fe$^{2+}$ Ion: $[Ar] 3d^6$ (Removed $4s^2$)
- Fe$^{3+}$ Ion: $[Ar] 3d^5$ (Removed $4s^2$ and one $3d$)
Photoelectron Spectroscopy (PES)
Photoelectron Spectroscopy is the experimental evidence that proves the shell model and electron configurations are correct.
How PES Works
High-energy photons (usually UV or X-rays) bombard a sample.
- If the photon energy ($h\nu$) is greater than the electron's binding energy ($BE$), the electron is ejected.
- The machine measures the Kinetic Energy ($KE$) of the ejected electron.
- We calculate Binding Energy using conservation of energy:
Think of $BE$ as the Ionization Energy required to remove that specific electron.
Interpreting the PES Spectrum
A PES graph plots Relative Number of Electrons (y-axis) vs. Binding Energy (x-axis).
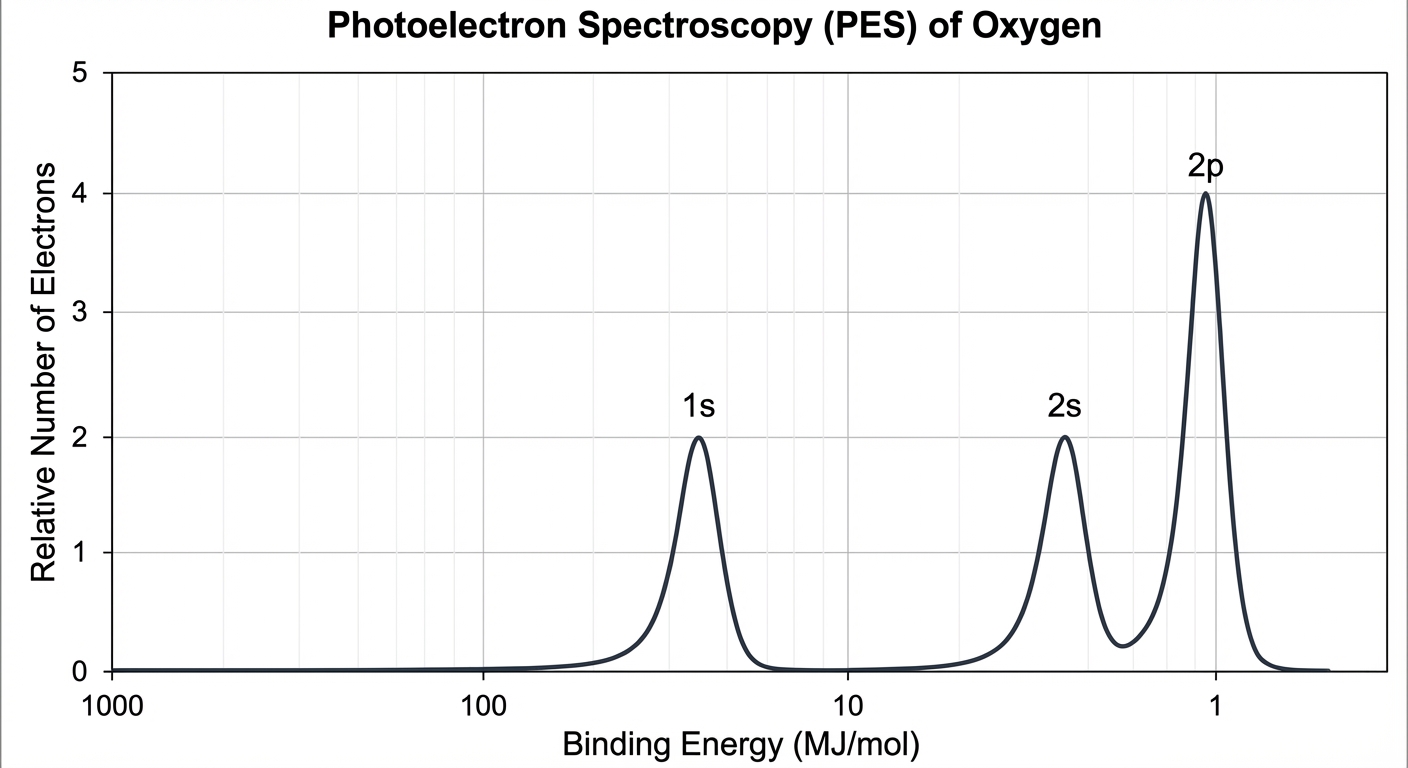
Reading the Axes
- X-Axis (Energy): Notice the scale usually goes high to low (left to right) or is logarithmic. High energy corresponds to electrons closer to the nucleus (Coulomb's Law: smaller $r$, stronger attraction).
- Y-Axis (Signal Intensity): The height of the peak is directly proportional to the number of electrons in that sublevel.
Analyzing Peaks
- Peak Position: Tells you the subshell ($1s$, $2s$, etc.). The peak with the highest binding energy (furthest left) is always the $1s$ orbital because it is closest to the nucleus.
- Peak Height: Tells you the ratio of electrons.
- If Peak A is height $h$ and Peak B is height $3h$, the ratio of electrons is 1:3 (e.g., $s^2$ vs $p^6$).
Example Analysis:
Imagine a graph with three peaks:
- Peak 1: Binding Energy 100 MJ/mol, Height = 2 units. (This is $1s^2$)
- Peak 2: Binding Energy 5 MJ/mol, Height = 2 units. (This is $2s^2$)
- Peak 3: Binding Energy 3 MJ/mol, Height = 1 unit. (This is $2p^1$)
- Total Electrons: $2+2+1 = 5$ (Boron).
Common Mistakes and Pitfalls
- Transition Metal Ion Error: Students frequently forget to remove $4s$ electrons before $3d$ electrons when forming transition metal cations.
- Wrong: Fe$^{2+}$ is $[Ar] 4s^2 3d^4$
- Right: Fe$^{2+}$ is $[Ar] 3d^6$
- Reading PES Backwards: Always check the x-axis on a PES graph. Usually, the highest energy (closest to nucleus) is on the left, but sometimes they flip it. Look for the numbers!
- Isoelectronic Confusion: Atoms/ions are isoelectronic if they have the same electron configuration (e.g., $F^-$ and $Ne$). Students often confuse this with having similar properties. They do not have the same properties because their nuclear charge (number of protons) is different.
- Coulombic Logic: When explaining why ionization energy trends occur, simply saying "shielding" or "radius" is often not enough to earn full points. You must explicitly reference Coulomb's Law: mention both the nuclear charge ($Q_1$) and the distance ($r$).