Unit 8 Comprehensive Guide: Acid-Base Equilibria and Titrations
1. Introduction to Acids and Bases
Definitions
Understanding the definitions of acids and bases is the foundation of this unit. While the Arrhenius definition is historical, Brønsted-Lowry is the primary focus of AP Chemistry.
- Arrhenius Acid: Produces $H^+$ (or $H_3O^+$) in water.
- Arrhenius Base: Produces $OH^-$ in water.
- Brønsted-Lowry Acid: A proton ($H^+$) donor.
- Brønsted-Lowry Base: A proton ($H^+$) acceptor.
Conjugate Acid-Base Pairs:
When an acid donates a proton, it becomes its conjugate base. When a base accepts a proton, it becomes its conjugate acid. These pairs differ by exactly one $H^+$.
The Autoionization of Water ($K_w$)
Water is amphoteric, meaning it can act as both an acid and a base. Pure water undergoes autoionization:
The equilibrium constant for this reaction is $K_w$:
Key Rules for $pH$ and $pOH$:
Temperature Dependence
$K_w$ acts like any equilibrium constant; it changes with temperature. The autoionization of water is endothermic ($bond breaking$).
- As Temp Increases: The reaction shifts right, $[H^+]$ increases, and pH decreases.
- Is it acidic? NO. Even though pH < 7 at high temperatures, $[H^+]$ still equals $[OH^-]$, so the water remains neutral.
2. Strength of Acids and Bases
Strong Acids and Bases
Strong acids and bases dissociate completely ($100\%$) in water. The reaction lies entirely to the right ($
ightarrow$), not at equilibrium ($
ightleftharpoons$).
The Big 6 Strong Acids (Memorize These):
- HCl (Hydrochloric acid)
- HBr (Hydrobromic acid)
- HI (Hydroiodic acid)
- $HNO_3$ (Nitric acid)
- $H2SO4$ (Sulfuric acid - only the first proton is strong)
- $HClO_4$ (Perchloric acid)
Strong Bases:
Group 1 and Group 2 hydroxides (e.g., $LiOH, NaOH, KOH, Ba(OH)2, Sr(OH)2$).
Weak Acids and Bases
Weak acids only partially dissociate. An equilibrium is established between the un-ionized acid and its ions. The extent of dissociation is quantified by $Ka$ (acid) or $Kb$ (base).
Formula for Weak Acid ($HA$):
- Larger $K_a$ = Stronger acid (more products, lower pH).
- Smaller $pK_a$ = Stronger acid.
Formula for Weak Base ($B$):
Relationship:
For a conjugate pair (e.g., $NH3$ and $NH4^+$):
Percent Ionization
- As the initial concentration of a weak acid decreases (becomes more dilute), the percent ionization increases (Le Chatelier’s Principle: dilution shifts equilibrium toward the side with more aqueous particles).
3. Molecular Structure and Acid Strength
Why are some acids strong and others weak? It depends on the bond identifying the ionizable hydrogen.
1. Binary Acids ($H-X$)
Acid strength depends on the bond strength between H and X.
- Trend: Acidity increases down a group.
- $HF < HCl < HBr < HI$
- Reasoning: As the atomic radius of X increases, the H-X bond becomes longer and weaker, making it easier for $H^+$ to break off.
- Note: HF is a weak acid because the H-F bond is extremely strong due to the small radius of Fluorine.
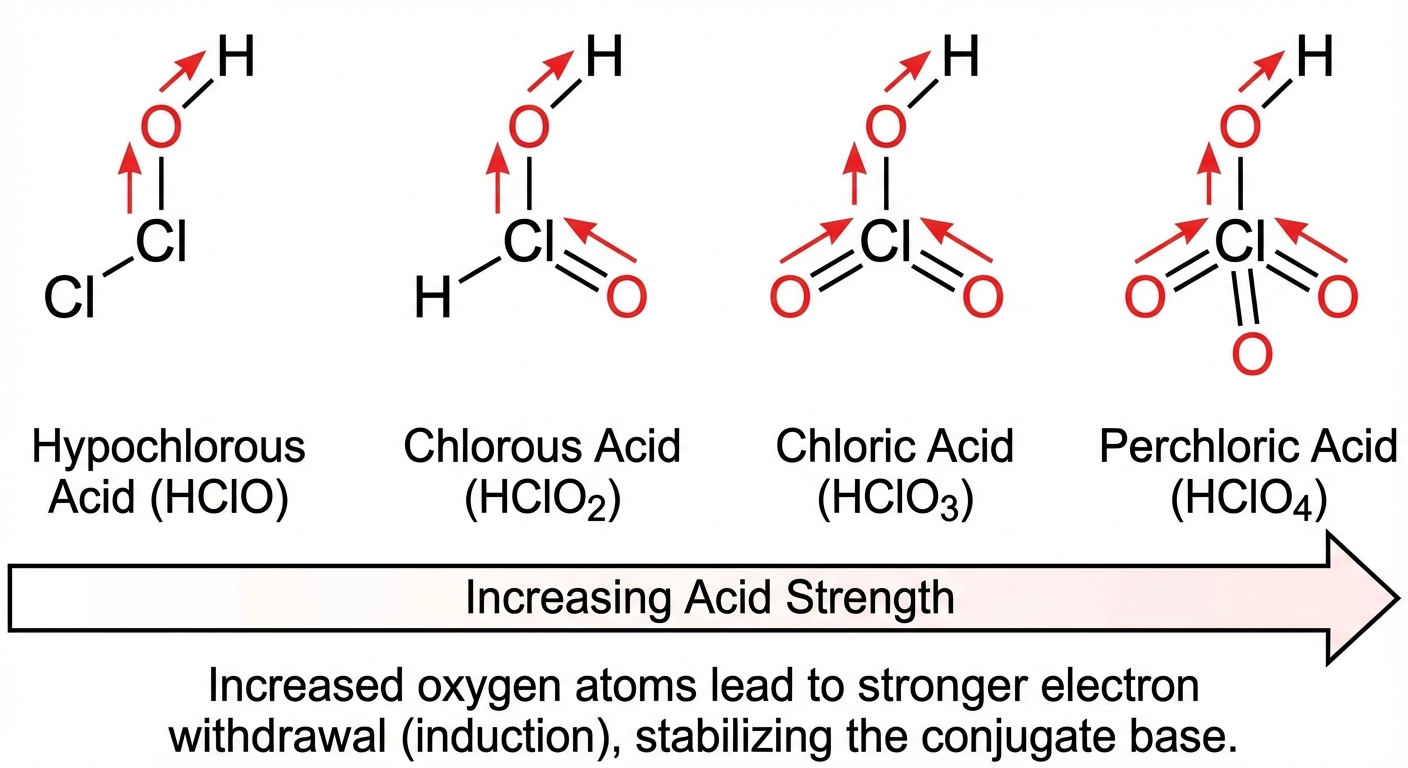
2. Oxyacids ($H-O-Z$)
Acid strength depends on the electronegativity of Z.
- Rule 1: If Z is more electronegative, it pulls electron density away from the O-H bond, weakening it and stabilizing the resulting anion.
- Example: $HOCl > HOI$ (Cl is more electronegative than I).
- Rule 2: Adding more Oxygen atoms increases acidity.
- Example: $HClO4 > HClO3 > HClO_2 > HClO$
- Reasoning: The extra oxygens draw electron density via induction, polarizing the O-H bond more and stabilizing the conjugate base via resonance.
3. Carboxylic Acids (-COOH)
The acidic proton is the one attached to the oxygen in the carboxyl group. The delocalization of electrons (resonance) over the two oxygen atoms in the conjugate base ($COO^-$) stabilizes the anion, driving dissociation.
4. Acid-Base Neutralization & Salts
When acids and bases react, the properties of the resulting salt depend on the strength of the parents.
Reaction Types
Strong Acid + Strong Base:
- Reaction: $H^+ + OH^- \rightarrow H_2O$
- Product: Water + Neutral Salt.
- pH at Equivalence: 7.00
Weak Acid + Strong Base:
- Reaction: $HA + OH^- \rightarrow A^- + H_2O$
- Note: The weak acid ($HA$) cannot be split into ions in the net ionic equation.
- Product: Water + Basic Salt.
- pH at Equivalence: > 7.00 (due to hydrolysis of $A^-$).
Strong Acid + Weak Base:
- Reaction: $H^+ + B \rightarrow HB^+$
- Product: Acidic Salt.
- pH at Equivalence: < 7.00 (due to hydrolysis of $HB^+$).
Hydrolysis of Salts (The nature of ions)
Do not assume salt water is neutral. Analyze the ions:
- Neutral Ions (Spectators): Conjugates of strong acids/bases ($Cl^-, NO_3^-, Na^+, K^+$). They do not react with water.
- Basic Ions: Conjugates of weak acids ($F^-, C2H3O_2^-$). They grab H from water, producing $OH^-$.
- Acidic Ions: Conjugates of weak bases ($NH_4^+$) or small, highly charged metal cations ($Al^{3+}, Fe^{3+}$). They release $H^+$.
5. Buffers
Definition
A buffer is a solution that resists changes in pH upon the addition of small amounts of strong acid or base. It must contain significant concentrations of both:
- A weak acid ($HA$)
- Its conjugate base ($A^-$)
How to make a buffer:
- Direct Mix: Mix acetic acid ($HC2H3O2$) and sodium acetate ($NaC2H3O2$).
- Partial Neutralization: React a weak acid with a limiting amount of Strong Base (e.g., 1 mole $HC2H3O_2 + 0.5$ mole $NaOH$). This converts half the acid into its conjugate base.
Henderson-Hasselbalch Equation
This equation allows for quick pH calculation of buffer solutions.
- Half-Equivalence Point: When $[A^-] = [HA]$, the log term becomes $\log(1) = 0$. Therefore, $pH = pK_a$.
Buffer Capacity
- Capacity: The amount of acid/base a buffer can absorb before pH changes drastically. Higher concentrations of HA and $A^-$ = Higher capacity.
- Ideal Range: Buffers differ by $\pm 1$ pH unit from their $pKa$. To choose a buffer for pH 5, pick an acid with $pKa \approx 5$.
6. Titrations
Titrations are controlled neutralization reactions used to determine the concentration of an unknown solution.
The Titration Curve
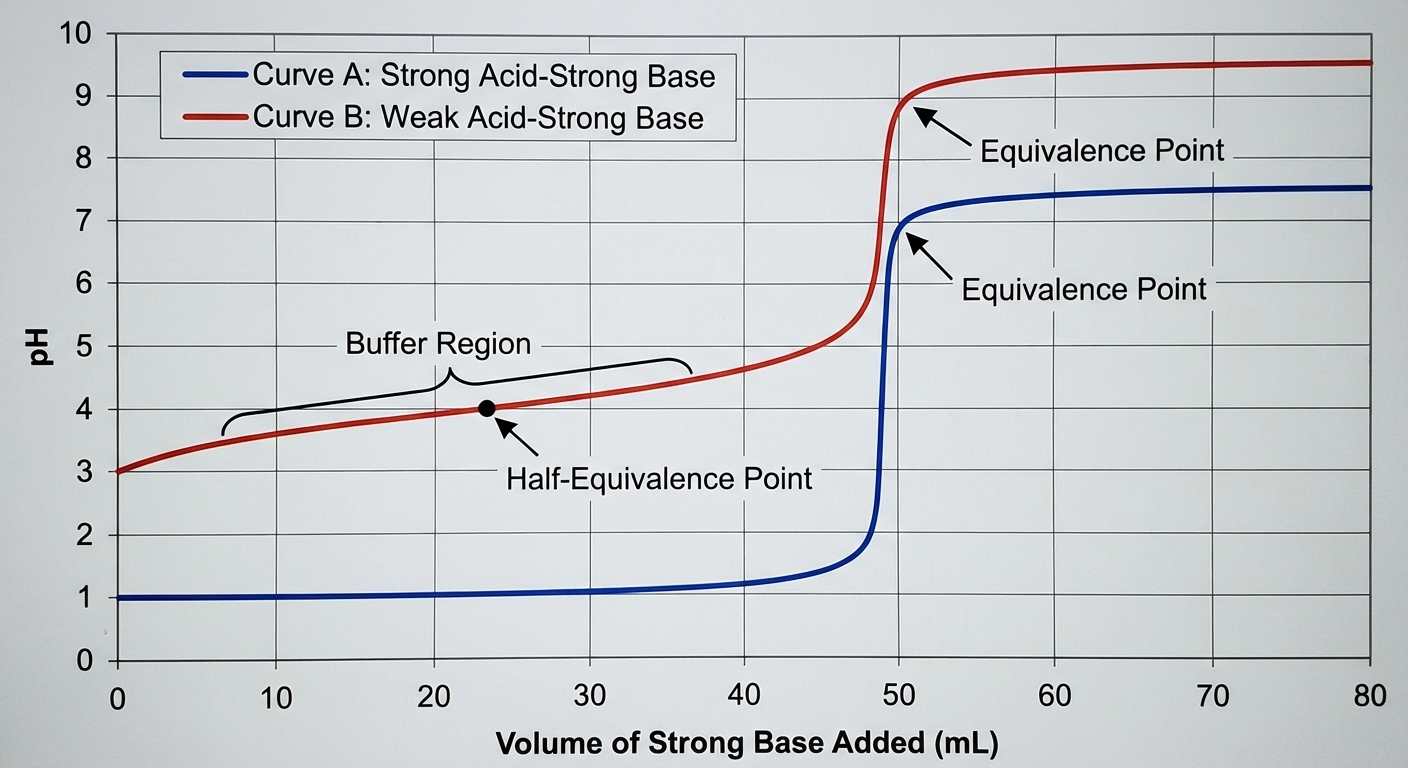
- Initial Region: pH depends solely on the analyte ($HA$). Use ICE table.
- Buffer Region: Between start and equivalence. The strong titrant turns weak analyte into conjugate. Use Henderson-Hasselbalch.
- Equivalence Point: Moles of acid = Moles of base. The solution contains only the conjugate salt. Use $Kb$ (or $Ka$) of the salt to find pH.
- Post-Equivalence: Excess strong titrant dominates. pH is determined strictly by the excess strong species.
| Feature | Strong Acid by Strong Base | Weak Acid by Strong Base |
|---|---|---|
| Initial pH | Very Low (~1) | Moderately Low (~3) |
| Shape | Flat, then vertical rise | "S" shape with buffer region |
| Equivalence pH | 7.00 | > 7.00 |
| Half-Equivalence | N/A | $pH = pK_a$ |
Indicators
Indicators are weak acids ($HIn$) that change color when they lose their proton ($In^-$).
- The color change occurs when $pH \approx pK_a$ of the indicator.
- Selection Rule: Choose an indicator whose $pK_a$ is close to the pH at the equivalence point of the titration.
Polyprotic Acids
Acids like $H3PO4$ have multiple equivalence points.
- $K{a1} >> K{a2} >> K_{a3}$ (It gets harder to remove protons as the ion becomes more negative).
- The curve looks like multiple weak acid waves connected.
7. Common Mistakes & Pitfalls
- Neutrality Misconception: Students often assume $pH = 7$ is always neutral. This is only true at $25^\circ$C. At $50^\circ$C, $K_w$ increases, so neutral pH might be 6.6.
- Weak x Strong Neutralization: When mixing a Weak Acid and Strong Base, students often try to use $M1V1 = M2V2$ for pH.
- Correction: You must use Stoichiometry (Moles) first to find the remaining species, then use Equilibrium (Henderson-Hasselbalch or ICE) to find pH.
- The $Ka$ vs $pKa$ confusion: Remember the signs. A high $Ka$ means a low $pKa$. Both indicate a stronger acid.
- Volume Changes: In titrations, remember that when you add titrant, the total volume increases. You must calculate new molarities ($M = mol / V_{total}$) before doing equilibrium calculations.
- Spectator Ions: Forgetting that ions like $Na^+$ and $Cl^-$ are spectators. They affect ionic strength but do not change pH.
- Common Ion Effect on Solubility: (Bridge to Unit 9) Adding a basic salt like $Mg(OH)_2$ to an acid will increase solubility because $H^+$ removes $OH^-$, shifting equilibrium right. Adding it to a base decreases solubility.