AP Chemistry: Intermolecular Forces and Properties
Intermolecular Forces (IMFs)
Intermolecular forces are the attractive forces between separate molecules (or atoms in noble gases), as opposed to intramolecular forces (chemical bonds) which exist within a molecule. Understanding these forces is crucial because they determine physical properties like boiling point, melting point, vapor pressure, and viscosity.
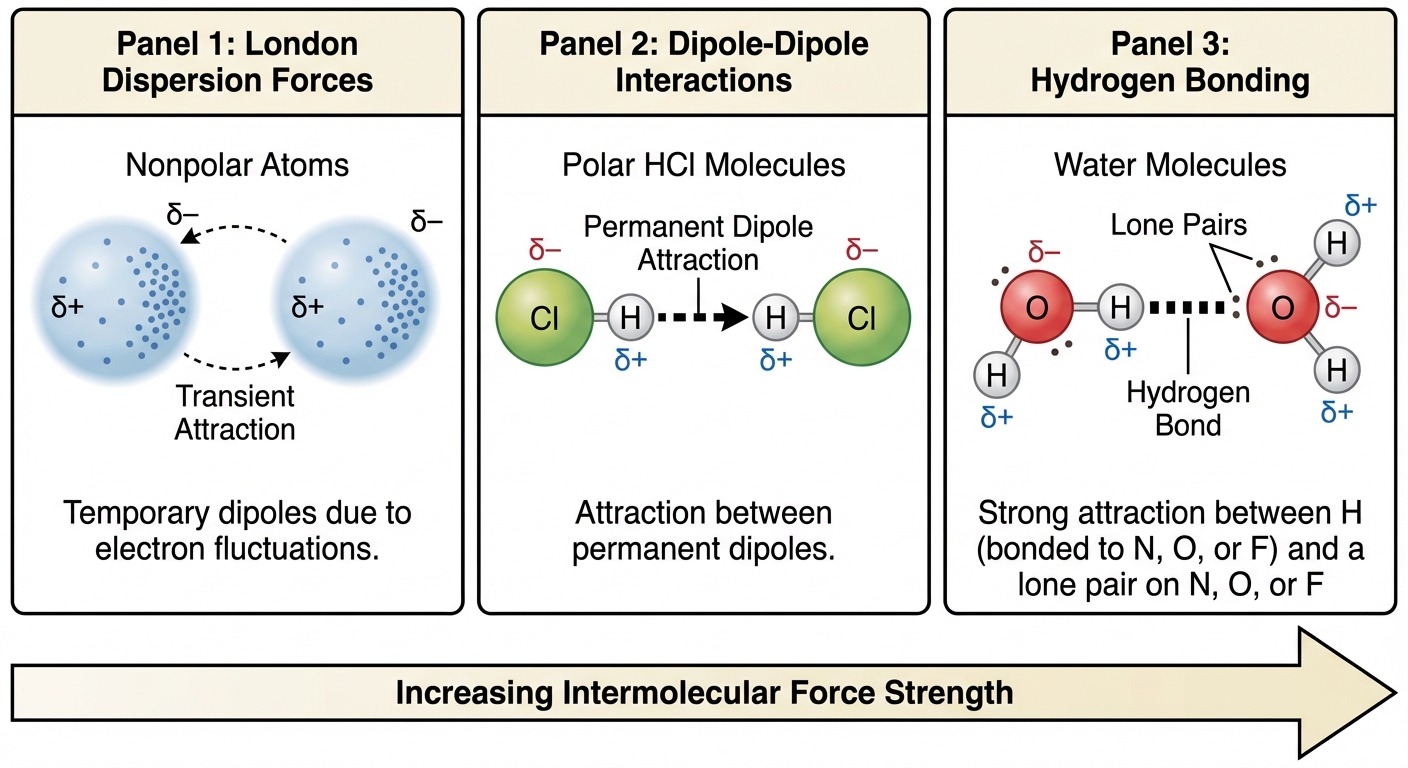
London Dispersion Forces (LDF)
London Dispersion Forces differ from other IMFs because they exist between all molecules and atoms, regardless of polarity.
- Mechanism: Electrons are in constant motion. At any given instant, the electron cloud can become unevenly distributed, creating a temporary, instantaneous dipole. This induces a dipole in a neighboring atom.
- Polarizability: The ease with which an electron cloud can be distorted is called polarizability.
- Larger electron clouds (more electrons) are more polarizable.
- Therefore, LDFs increase in strength as the molar mass (and number of electrons) increases.
- Shape Factor: Long, cylindrical molecules have stronger LDFs than spherical molecules of similar mass because they have more surface area for contact.
Dipole-Dipole Forces
These occur between molecules that have permanent dipoles (polar molecules).
- Mechanism: The partially positive end ($͇+$) of one molecule is attracted to the partially negative end ($͇-$) of another.
- Strength: Generally stronger than LDFs for molecules of similar size. However, a very large nonpolar molecule (strong LDFs) can have a higher boiling point than a small polar molecule.
Hydrogen Bonding
Hydrogen bonding is not a true chemical bond; it is an unusually strong specific type of dipole-dipole attraction.
- Criteria: Occurs ONLY when Hydrogen is covalently bonded to Nitrogen, Oxygen, or Fluorine (N, O, F).
- Why? N, O, and F are highly electronegative and small. This creates a vary large dipole and exposes the hydrogen nucleus (proton), allowing very close approach and strong attraction to a lone pair on a neighboring N, O, or F.
- Consequences: High boiling points (e.g., $H2O$, $HF$, $NH3$) compared to homologs like $H2S$ or $PH3$.
Ion-Dipole Forces
These occur when an ionic compound dissolves in a polar solvent.
- Mechanism: The cations are attracted to the negative pole of the solvent, and anions are attracted to the positive pole.
- Importance: This force is stronger than Hydrogen bonding and explains why ionic salts dissolve in water.
Comparison of IMFs
Generally, the strength hierarchy is:
LDF < Dipole\text{-}Dipole < Hydrogen\ Bonding < Ion\text{-}Dipole < Covalent/Ionic\ Bonds
Note: This hierarchy assumes molecules of comparable size. A large molecule with only LDFs can have stronger total attraction than a tiny molecule with H-bonds.
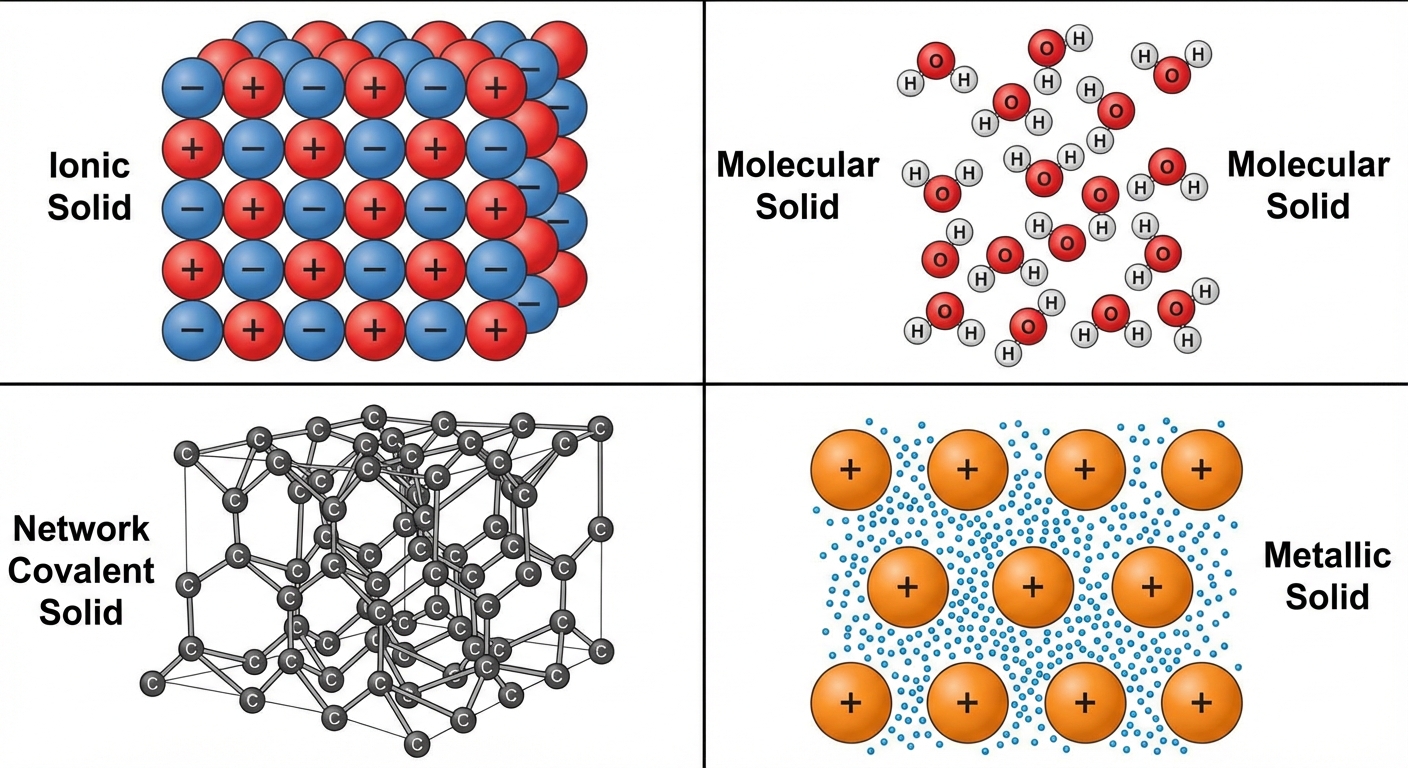
Properties of Solids
Solids are classified by the types of particles and the forces holding them together. This classification predicts properties like conductivity, malleability, and melting point.
1. Ionic Solids
- Particles: Ions (cations and anions).
- Force: Coulombic attraction (ionic bonds) in a 3D lattice.
- Properties:
- High melting/boiling points.
- Brittle (repulsion occurs when layers slide).
- Poor conductors as solids; good conductors when molten or dissolved (ions are free to move).
2. Molecular Solids
- Particles: Distinct neutral molecules.
- Force: Intermolecular Forces (LDF, Dipole, H-bonds).
- Properties:
- Low melting points (easy to overcome IMFs).
- Poor conductors (no free charges).
- Soft/brittle.
- Examples: Ice ($H2O$), Dry Ice ($CO2$), Sugar ($C{12}H{22}O_{11}$).
3. Network Covalent Solids
- Particles: Atoms connected in a vast 2D or 3D network.
- Force: Covalent bonds (intramolecular).
- Properties:
- Extremely high melting points.
- Very hard.
- Generally poor conductors (Exception: Graphite forms sheets with delocalized electrons).
- Examples: Diamond ($C$), Quartz ($SiO_2$), Silicon Carbide ($SiC$).
4. Metallic Solids
- Particles: Metal cations in a "sea of delocalized electrons."
- Force: Metallic bonding.
- Properties:
- Malleable and ductile (bonding is non-directional).
- Excellent thermal and electrical conductors.
- Wide range of melting points.
- Alloys: Mixtures of metals. Interstitial alloys form when small atoms fit between metal atoms (e.g., Steel); Substitutional alloys form when atoms of similar size replace metal atoms (e.g., Brass).
Liquids and Valance Properties
The strength of IMFs directly influences physical properties of liquids.
- Vapor Pressure: The pressure exerted by a gas in equilibrium with its liquid phase. Stronger IMFs = Lower Vapor Pressure (harder for molecules to escape into gas).
- Boiling Point: The temperature where vapor pressure equals atmospheric pressure. Stronger IMFs = Higher Boiling Point.
- Viscosity: Resistance to flow. Stronger IMFs = Higher Viscosity.
- Surface Tension: Energy required to increase surface area. Stronger IMFs = Higher Surface Tension.
The Gas Laws and KMT
Kinetic Molecular Theory (KMT)
Ideal gases are theoretical, but real gases approximate them under certain conditions. KMT assumptions are:
- Gas particles are in continuous, random motion.
- Volume of the particles is negligible compared to the container volume.
- No forces of attraction or repulsion exist between particles.
- Collisions are completely elastic (no net loss of kinetic energy).
- Average Kinetic Energy depends only on Temperature ($KE_{avg} \propto T$).
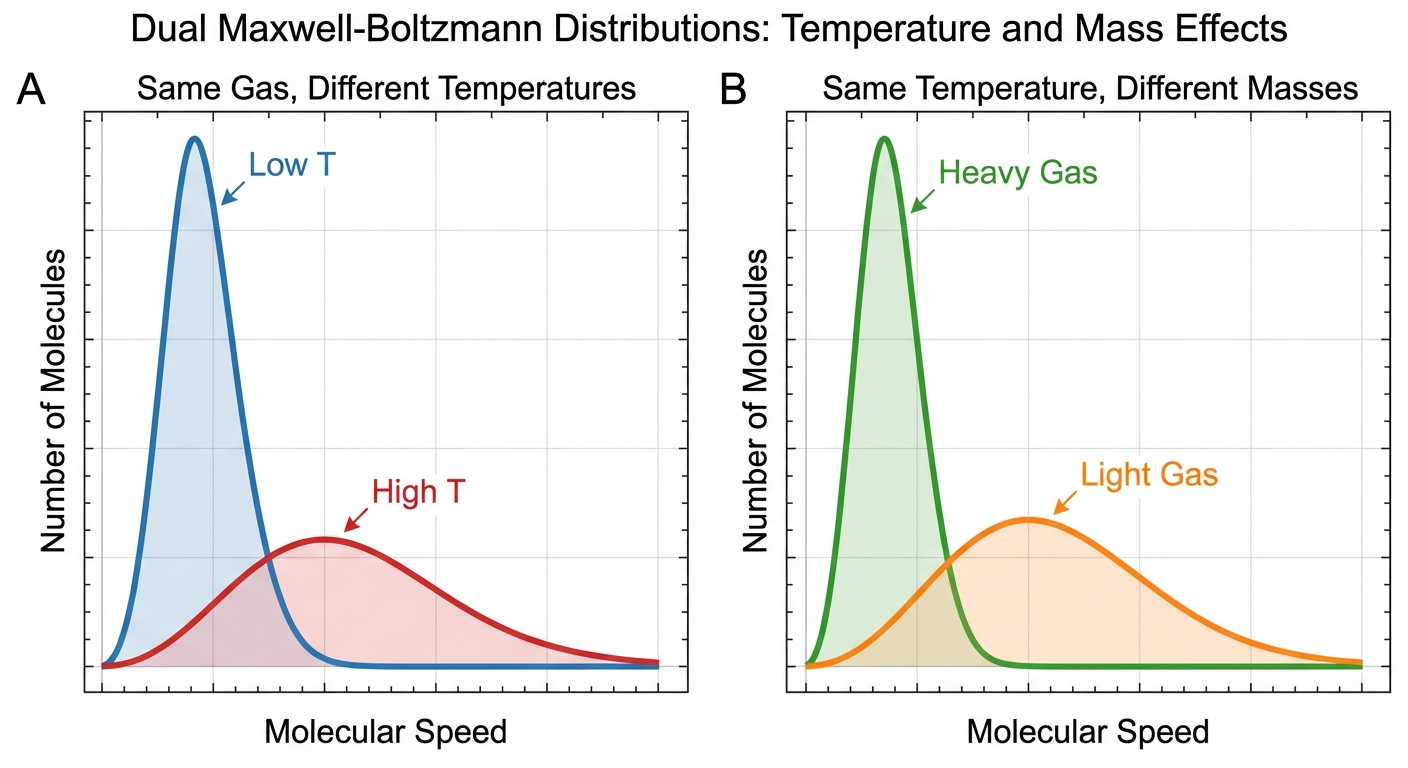
Maxwell-Boltzmann Distributions
These graphs show the distribution of particle speeds in a gas sample.
- Effect of Temperature: As T increases, the curve flattens and shifts right. A greater fraction of molecules have high speeds.
- Effect of Molar Mass: At a constant T, heavier gases move slower. Lighter gases have a broader distribution shifted to the right.
Ideal Gas Equation
- $P$ = Pressure (atm, torr, Pa)
- $V$ = Volume (L)
- $n$ = Moles (mol)
- $T$ = Temperature (Must be in Kelvin)
- $R$ = Ideal Gas Constant ($0.08206\ L\cdot atm/mol\cdot K$)
Dalton’s Law of Partial Pressures
Where $XA$ is the mole fraction: $XA = \frac{nA}{n_{total}}$.
Deviations from Ideal Behavior
Real gases deviate from the Ideal Gas Law when KMT assumptions break down:
- High Pressure: Particles are forced close together; particle volume becomes significant.
- Low Temperature: Particles move slowly; IMFs become significant (attractions pull particles together, lowering observed pressure).
Key Rule: Gases are most ideal at High Temperature and Low Pressure.
Solutions and Mixtures
Molarity and Preparation
Concentration is typically measured in Molarity ($M$):
To prepare a solution, dissolve the solute in less water than the final volume, then dilute to the mark on the volumetric flask.
Solubility and "Like Dissolves Like"
- Polar solvents dissolve polar or ionic solutes.
- Nonpolar solvents dissolve nonpolar solutes.
- Substances with both polar and nonpolar parts (like alcohols) vary in solubility depending on which part dominates the structure.
Separation Techniques
Separating mixtures relies on differences in intermolecular properties.
- Distillation: Separates liquids based on differences in boiling points (and thus IMF strength).
- Chromatography (Paper/Thin Layer): Separates interacting components based on polarity.
- Stationary Phase: Usually polar (paper).
- Mobile Phase: Solvent (can be polar or nonpolar).
- If the solvent is nonpolar, the nonpolar components of the mixture travel extensively (high $Rf$ value), while polar components "stick" to the polar paper (low $Rf$ value).

Spectroscopy and Light
Matter interacts with electromagnetic radiation in specific ways depending on the energy of the photons.
Types of Transitions (Memorize These interactions)
- Microwave Radiation: Causes molecular rotation.
- Infrared (IR) Radiation: Causes molecular vibration (useful for identifying functional bond types).
- UV/Vis Radiation: Causes electronic transitions (electrons jump to higher energy levels).
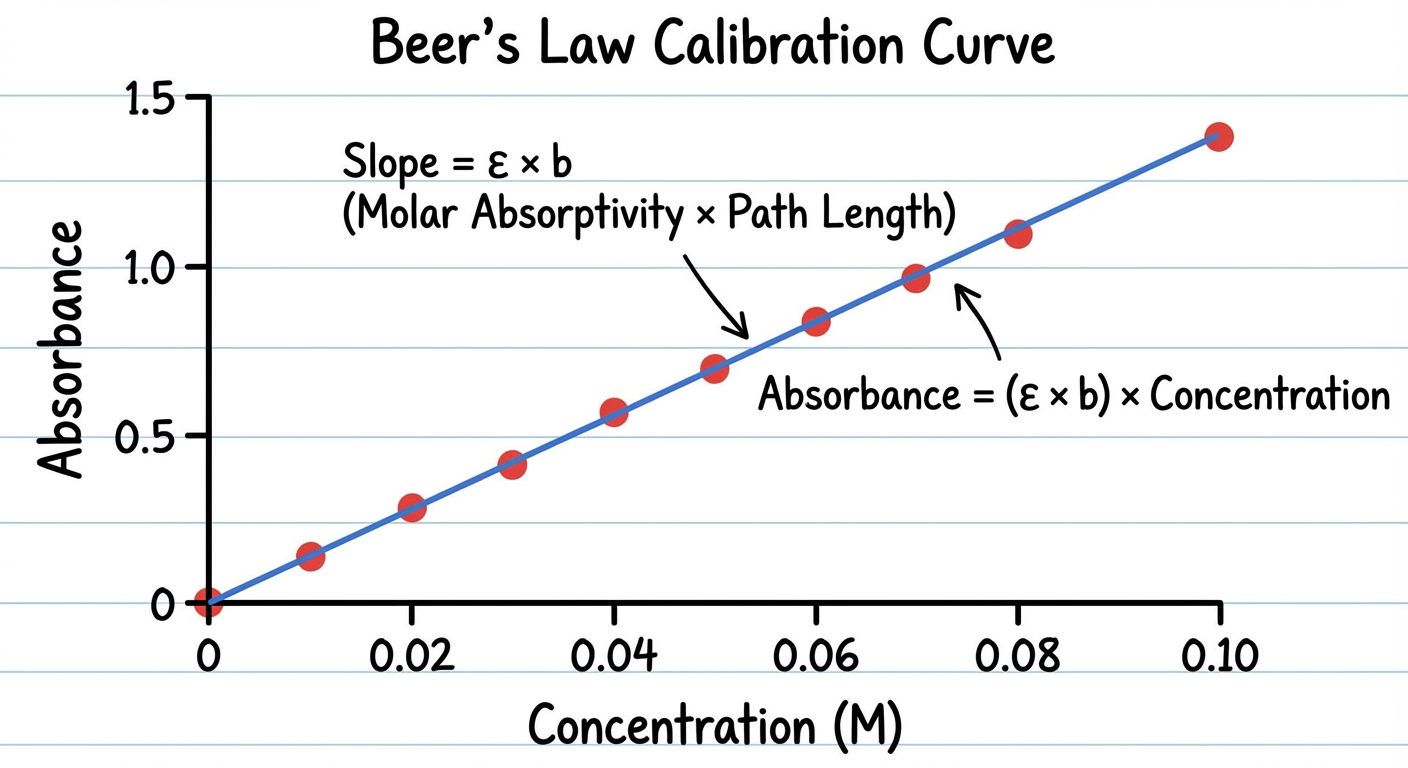
Beer-Lambert Law
Used to calculate the concentration of a colored solution based on how much light it absorbs (using a spectrophotometer).
- $A$ = Absorbance (no units)
- $\epsilon$ = Molar absorptivity constant ($M^{-1}cm^{-1}$)
- $b$ = Path length (width of cuvette, usually 1 cm)
- $c$ = Concentration ($M$)
Relationship: Absorbance is directly proportional to Concentration. If you plot Absorbance ($y$) vs. Concentration ($x$), the slope is $\epsilon b$.
Common Mistakes & Pitfalls
- Confusing IMF strength with Bond Strength: Covalent bonds are strong; IMFs are weak. Boiling water breaks IMFs (H-bonds), NOT covalent bonds (you don't get H and O gas).
- The "Largest Molecule" Trap: Students often assume H-bonding naturally makes a molecule have the highest boiling point. A massive nonpolar molecule (like $C{20}H{42}$) often boils higher than small H-bonded molecules (like $H_2O$) because the LDFs in the large molecule sum up to a greater total force.
- Phase Change Temp: Temperature does not change during a phase change (plateaus on a heating curve). Energy goes into breaking IMFs, not increasing kinetic energy.
- Network Solid vs Molecular Solid: Silicon dioxide ($SiO2$) is a network solid (sand/quartz) with a very high MP. Carbon dioxide ($CO2$) is a discrete molecule with a low MP. Do not confuse them just because they look similar in formula.
- Gas Law Units: Always use Kelvin for temperature. For $R = 0.08206$, Pressure must be atm and Volume must be Liters.
- Breaking Bonds in Solutions: Dissolving is not "melting." Don't say salt melts in water; it dissociates.