AP Chemistry Unit 6: Thermodynamics Study Notes
Fundamentals of Energy and Heat
Kinetic Energy, Temperature, and Heat
Thermodynamics is the study of energy and its interconversions. In AP Chemistry, we focus primarily on the First Law of Thermodynamics, which states that energy cannot be created or destroyed, only transferred or transformed.
- Temperature ($T$): A measure of the average kinetic energy of the particles in a substance. It is independent of the amount of substance.
- Equation: $KE_{avg} \propto T$ (in Kelvin)
- Thermal Energy: The total kinetic energy associated with the random motion of atoms and molecules. It depends on the amount of substance (mass) and the temperature.
- Heat ($q$): The transfer of energy between two substances due to a temperature difference. Heat always flows spontaneously from the hotter object to the colder object until thermal equilibrium (equal temperatures) is reached.
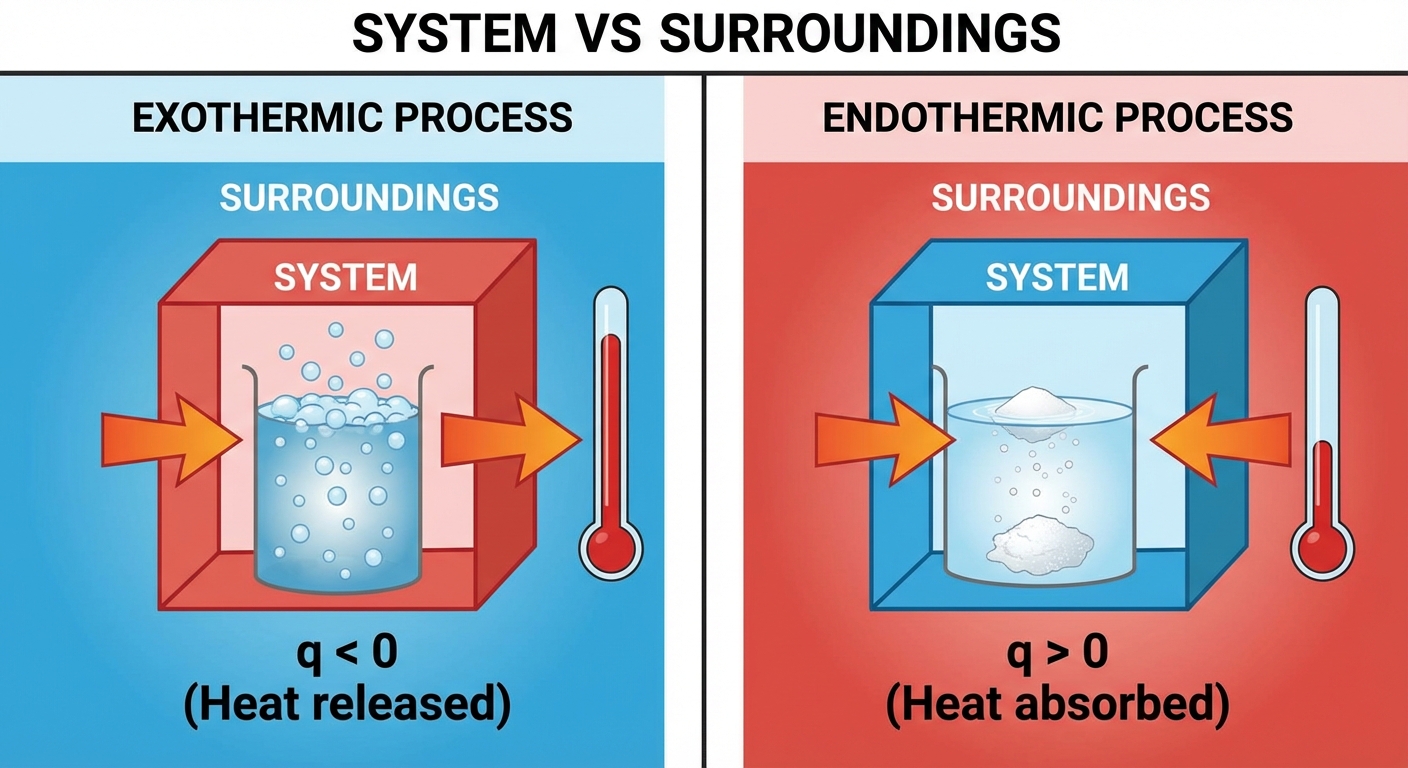
System vs. Surroundings
To analyze energy changes, we must define the universe in two parts:
- System: The specific part of the universe being studied (usually the chemical reaction or the specific chemicals in a beaker).
- Surroundings: Everything else in the universe (the water solvent, the beaker, the air, the thermometer).
Work and Energy Transfer
The change in internal energy ($\Delta E$) of a system is the sum of heat ($q$) and work ($w$):
- Work ($w$): In chemistry, this is usually pressure-volume work due to gas expansion or compression ($w = -P\Delta V$).
- Gas expands: System does work on surroundings ($w$ is negative).
- Gas compresses: Surroundings do work on system ($w$ is positive).
Endothermic and Exothermic Processes
The most critical concept in Unit 6 is the direction of energy flow.
Exothermic Processes ($q < 0$)
Energy is transferred from the system to the surroundings.
- Sign Convention: $\Delta H$ is negative ($-$).
- Observation: The surroundings (e.g., the reaction vessel water) gain energy, so the temperature measured by a thermometer increases.
- Bonding: Energy is released when bonds are formed. In exothermic reactions, the bonds formed in products are stronger/more stable than the bonds broken in reactants.
- Examples: Combustion, freezing, condensation, deposition.
Endothermic Processes ($q > 0$)
Energy is absorbed by the system from the surroundings.
- Sign Convention: $\Delta H$ is positive ($+$).
- Observation: The surroundings lose energy to the system, so the temperature measured by a thermometer decreases.
- Bonding: Energy is absorbed to break bonds. In endothermic reactions, the bonds in the products are weaker/less stable than the bonds in the reactants.
- Examples: Photosynthesis, melting, boiling, sublimation.
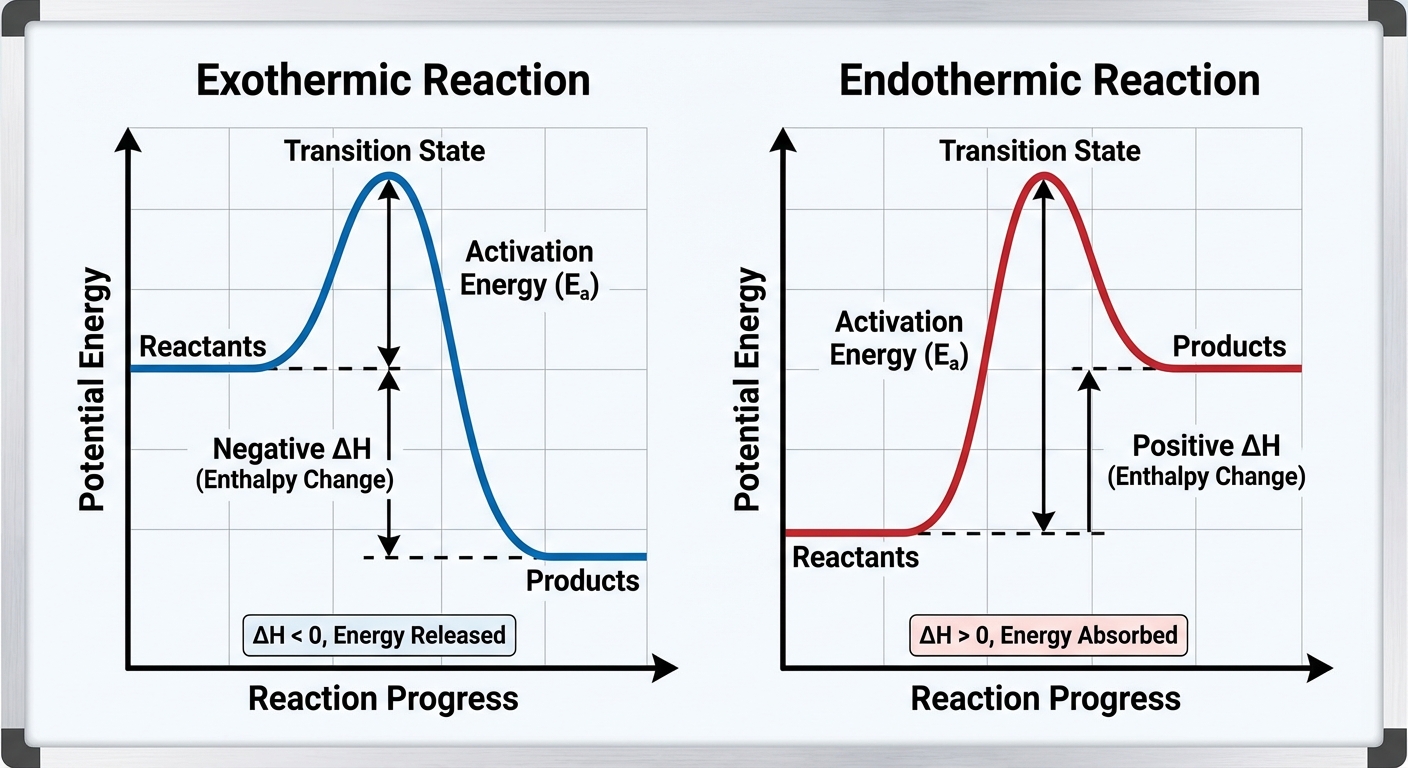
Energy Diagrams and Reaction Profiles
Potential energy diagrams visualize the energy changes during a reaction path.
Key Components of an Energy Diagram
- Reactants & Products: Plotted on the y-axis (Potential Energy).
- Activation Energy ($E_a$): The minimum energy required to initiate the reaction. It is the energy difference between the reactants and the activated complex (transition state) at the peak.
- Enthalpy Change ($\Delta H$): The difference in potential energy between products and reactants.
Comparing Profiles
- Exothermic: Products are at a lower energy level than reactants. Energy is released.
- Endothermic: Products are at a higher energy level than reactants. Energy is absorbed.
Effect of Catalysts
A catalyst speeds up a reaction by providing an alternate reaction pathway with a lower activation energy ($E_a$).
- The catalyst stabilizes the transition state.
- Crucial Note: A catalyst does not change the initial energy of reactants, the final energy of products, or the $\Delta H$ of the reaction.
Calorimetry and Heat Capacity
Calorimetry is the experimental measurement of heat flow.
Specific Heat Capacity ($c$ or $C_p$)
The amount of heat required to raise the temperature of 1 gram of a substance by 1°C (or 1 K).
- High specific heat: Substance resists temperature change (like water).
- Low specific heat: Substance heats up/cools down quickly (like metals).
The Heat Equation
To calculate the heat absorbed or released when temperature changes (and no phase change occurs):
- $q$ = heat (Joules)
- $m$ = mass (grams)
- $c$ = specific heat capacity ($J/(g\cdot ^\circ C)$)
- $\Delta T$ = $T{final} - T{initial}$
Conservation of Energy in Calorimetry
In a perfectly insulated calorimeter (like a coffee-cup calorimeter), heat lost by the reaction equals heat gained by the solution (or vice versa).
Common Scenario: Hot Metal into Cold Water
Study Tip: Always double-check your signs for $\Delta T$. If a substance cools down, $\Delta T$ is negative, resulting in a negative $q$.
Thermodynamics of Phase Changes
Phase changes involve changes in potential energy (intermolecular distance), not kinetic energy (speed). Therefore, temperature remains constant during a phase change.
Enthalpy of Fusion and Vaporization
To calculate heat during a phase change, we cannot use $mc\Delta T$ because $\Delta T = 0$. Instead, we use enthalpy values.
- Melting/Freezing: Uses Enthalpy of Fusion ($\Delta H_{fus}$).
- (where $n$ is moles)
- Boiling/Condensing: Uses Enthalpy of Vaporization ($\Delta H_{vap}$).
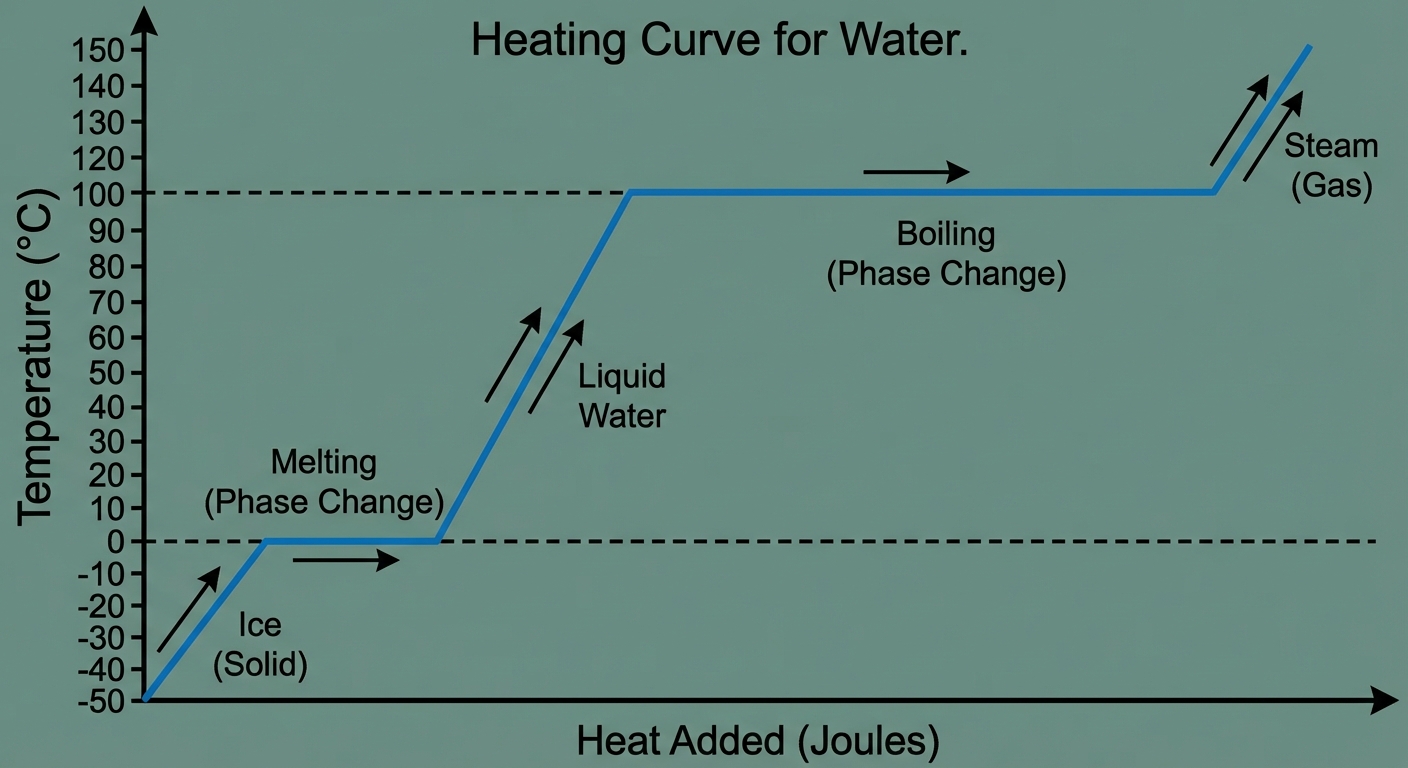
Heating Curves
A graph of Temperature vs. Heat Added (or Time).
- Sloped lines: Single phase heating up. Kinetic energy increases. Use $q=mc\Delta T$.
- Flat Plateaus: Phase change occurring. Potential energy increases (overcoming IMFs). Kinetic energy is constant. Use $q=n\Delta H$.
Enthalpy of Reaction ($\Delta H_{rxn}$)
Enthalpy ($H$) is a state function, composed of internal energy, pressure, and volume. We generally only measure the change in enthalpy.
There are three primary methods to calculate $\Delta H_{rxn}$ on the AP exam:
1. Bond Enthalpies
Bond breaking is endothermic (+); Bond forming is exothermic (-).
Formula:
Note: This is effectively Reactants minus Products.Example Calculation: Using bond energies to find $\Delta H$ for $2H2 + O2 \rightarrow 2H_2O$.
- Bonds Broken: 2 H-H bonds, 1 O=O bond.
- Bonds Formed: 4 O-H bonds.
- $\Delta H = [2(436) + 498] - [4(463)] = -482 \text{ kJ/mol}$ (Exothermic)
2. Standard Enthalpy of Formation ($\Delta H_f^\circ$)
The enthalpy change when one mole of a compound is formed from its elements in their standard states (1 atm, 25°C).
Important Rule: The $\Delta Hf^\circ$ of any pure element in its standard state (e.g., $O2(g)$, $Fe(s)$, $Br_2(l)$) is ZERO.
Formula:
Note: This is Products minus Reactants.
3. Hess's Law
If a reaction is carried out in a series of steps, $\Delta H$ for the overall reaction is the sum of the enthalpy changes for the individual steps.
Rules for Manipulation:
- Reverse Equation: Flip the sign of $\Delta H$. ($+ \rightarrow -$)
- Multiply Coefficients: Multiply $\Delta H$ by the same factor.
- Add Equations: Add the $\Delta H$ values together.
Example:
Target: $A \rightarrow B$
Given:
1) $A \rightarrow C$ ($\Delta H = 50$)
2) $B \rightarrow C$ ($\Delta H = 20$)
Strategy: Keep eq 1 as is. Flip eq 2 ($C \rightarrow B, \Delta H = -20$).
Add them: $(A \rightarrow C) + (C \rightarrow B) \Rightarrow A \rightarrow B$
$\Delta H_{total} = 50 + (-20) = 30$.
Enthalpy of Solution
When a salt dissolves in water, the process involves three energy steps:
- Solute Separation: Breaking ionic bonds in the lattice. (Endothermic, requires Energy).
- Solvent Separation: Overcoming IMFs (H-bonds) in water to make room for ions. (Endothermic).
- Hydration (Mixing): Attraction between ions and water dipoles. (Exothermic, releases Energy).
If the energy released in step 3 is greater than the energy required in steps 1 & 2, the dissolving is exothermic (pack heats up). If less, it is endothermic (pack cools down).
Common Mistakes & Pitfalls
- Sign Errors: This is the most common mistake. Remember: Exothermic = Negative (-); Endothermic = Positive (+).
- Equation Confusion:
- Using Bond Energies: It is Reactants - Products.
- Using Enthalpies of Formation: It is Products - Reactants.
- Units Mismatch: Specific heat is often in Joules (J), while $\Delta H$ is often in Kilojoules (kJ). You must convert to the same unit before adding or solving.
- Temperature in Phase Change: Students often try to use $q=mc\Delta T$ for melting or boiling. Remember, $\Delta T=0$ during a phase change; use heat of fusion/vaporization instead.
- Coefficients in Hess's Law: Forgetting to multiply the $\Delta H$ value when you multiply the coefficients of a reaction step.
- Standard States: Forgetting that diatomic elements (like $O2, N2, Cl2$) have a $\Delta Hf^\circ$ of zero. The problem usually won't give you this value; you are expected to know it.