AP Biology Unit 3: Cellular Energetics Comprehensive Guide
Unit 3: Cellular Energetics
This unit explores how living systems capture, store, and use energy. From the specificity of enzymes to the complex pathways of photosynthesis and cellular respiration, understanding bioenergetics is fundamental to biology.
Enzyme Structure and Function
Enzymes are biological macromolecules (typically proteins) that act as catalysts, meaning they speed up chemical reactions without being consumed by the reaction.
Structure and Specificity
- Active Site: A specific region on the enzyme where the substrate binds. The shape and chemical properties (charge, hydrophobicity) of the active site must match the substrate.
- Substrate: The reactant molecule that the enzyme acts upon.
- Enzyme-Substrate Complex: The temporary association formed when the substrate binds to the active site.
- Induced Fit Model: Unlike a rigid "lock and key," the enzyme changes shape slightly when binding to the substrate, tightening the fit to catalyze the reaction more effectively.
Catalysis Mechanism
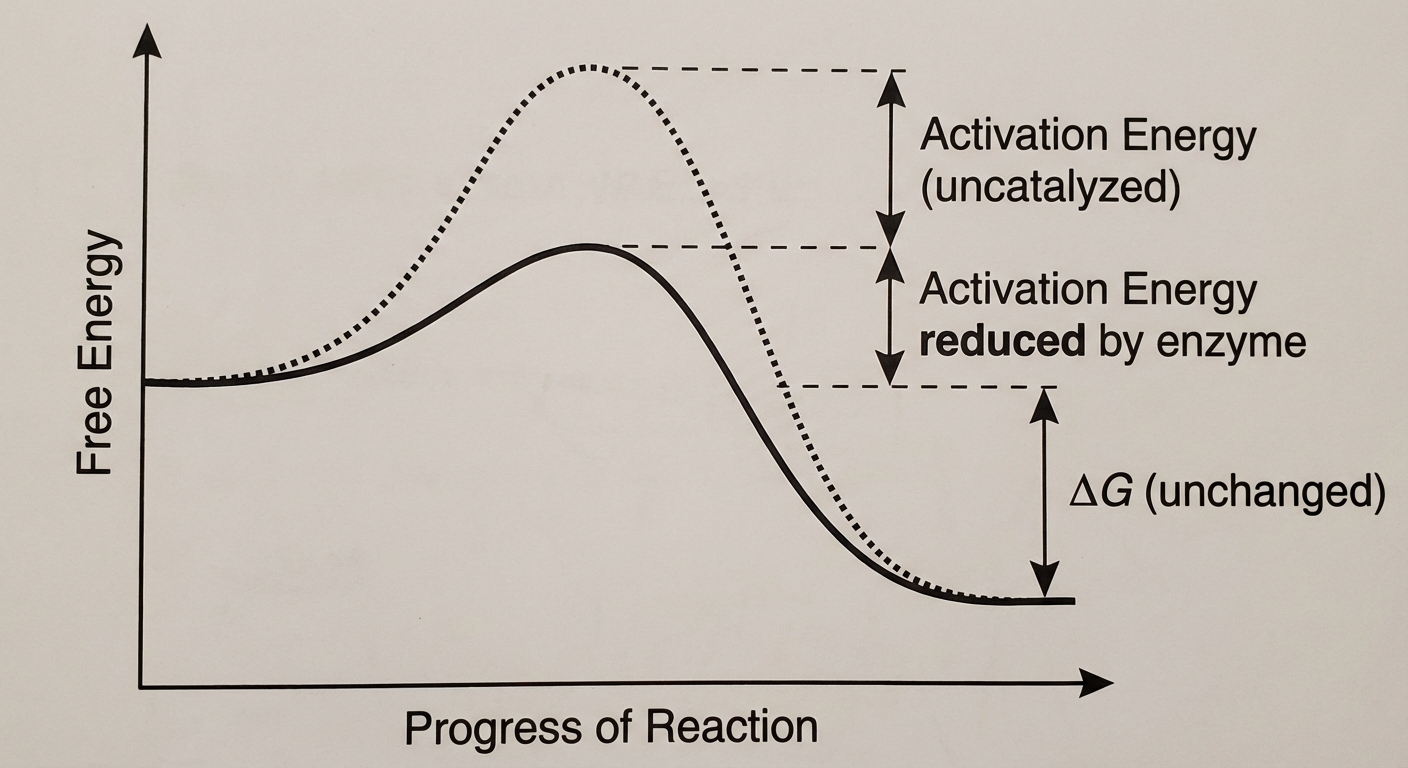
Enzymes speed up reactions by lowering the Activation Energy ($E_a$).
- $E_a$ is the initial energy investment required to start a reaction (to break bonds of reactants).
- Key Concept: Enzymes do NOT change the free energy ($\Delta G$) of the reaction. An exergonic reaction remains exergonic; the enzyme just makes it happen faster.
Environmental Impacts on Enzyme Function
An enzyme's structure (tertiary shape) determines its function. Changing the environment can alter this structure.
- Temperature:
- Optimal Range: Collisions increase with heat, increasing rate.
- Too High: Thermal agitation disrupts hydrogen bonds, causing denaturation (loss of shape and function).
- Too Low: Rate decreases due to fewer molecular collisions (usually essentially reversible).
- pH:
- Each enzyme has an optimal pH (e.g., Stomach pepsin: pH 2; Intestinal trypsin: pH 8).
- Deviations disrupt hydrogen bond networks in the active site, leading to denaturation.
- Substrate Concentration:
- Increasing substrate increases rate until the Saturation Point.
- Saturation: All active sites are occupied. Adding more substrate will not increase the rate further unless more enzyme is added.
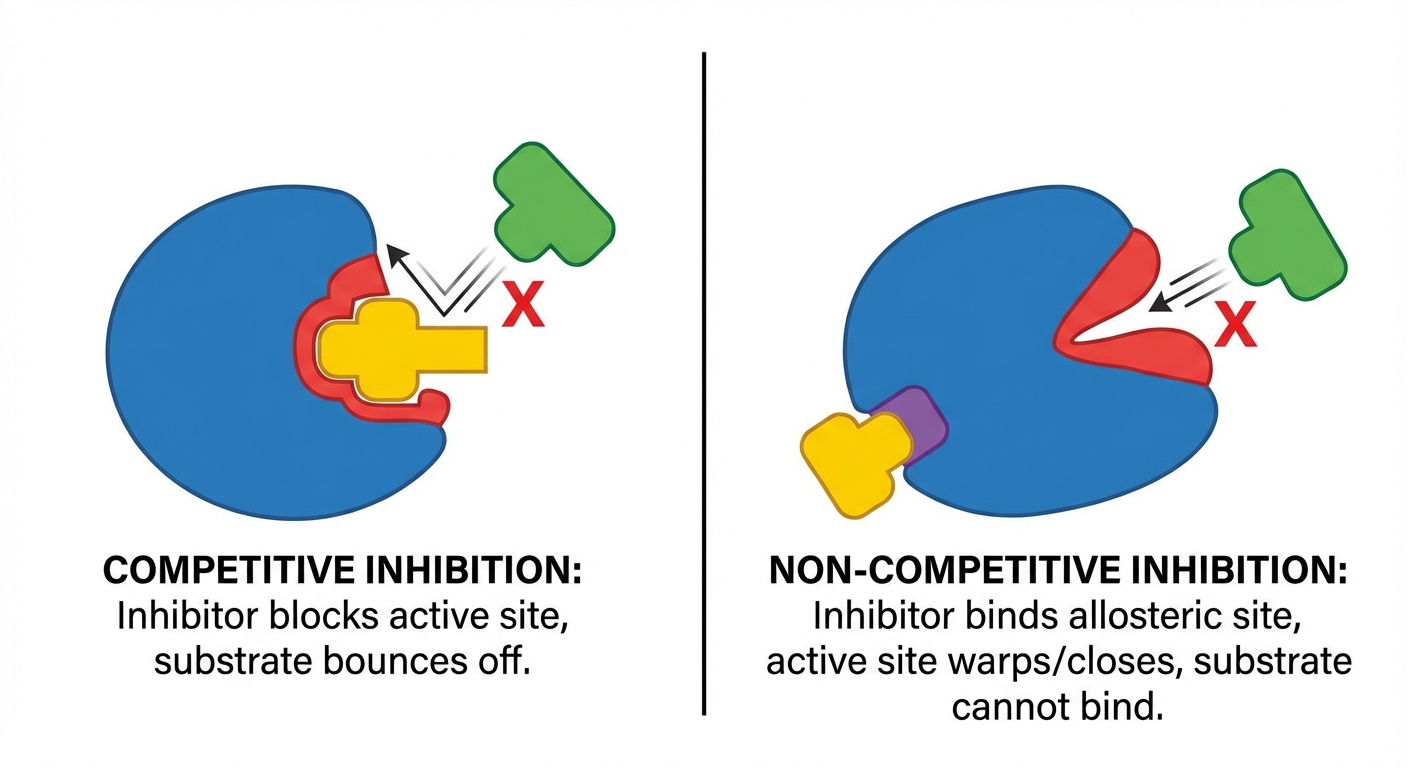
Enzyme Regulation & Inhibition
Cells must control when and where enzymes function.
| Type | Mechanism | Effect of Adding More Substrate |
|---|---|---|
| Competitive Inhibition | Inhibitor binds to the active site, blocking the substrate. | Overcome: Yes. If you flood the system with substrate, it outcompetes the inhibitor. |
| Non-competitive Inhibition | Inhibitor binds to an allosteric site (a site other than the active site). This changes the shape of the enzyme, making the active site non-functional. | Overcome: No. Adding more substrate does not fix the broken enzyme shape. |
Thermodynamics and Energy Coupling
Laws of Thermodynamics
- First Law: Energy cannot be created or destroyed, only transformed (e.g., Light energy $\to$ Chemical energy).
- Second Law: Every energy transfer increases the entropy (disorder) of the universe. Cells require constant energy input to maintain their highly ordered state against this tide of entropy.
Types of Reactions
- Exergonic: Releases energy. Products have less free energy than reactants ($\Delta G < 0$). Spontaneous.
- Example: Cellular Respiration, ATP hydrolysis.
- Endergonic: Requires energy input. Products have more free energy ($\Delta G > 0$). Non-spontaneous.
- Example: Photosynthesis, Protein synthesis.
Energy Coupling
Cells manage energy resources by interaction coupling: using the energy formulated from an exergonic process to drive an endergonic one.
The Role of ATP (Adenosine Triphosphate):
- The bond between the 2nd and 3rd phosphate is unstable and holds high potential energy caused by the repulsion of negative charges.
- Hydrolysis of this bond releases energy used to power cellular work.
Photosynthesis
Definition: A process capturing light energy to convert carbon dioxide and water into carbohydrates and oxygen.
Evolutionary History: First evolved in prokaryotes (cyanobacteria). This oxygenated the early Earth's atmosphere, allowing for the evolution of aerobic life.
General Formula:
Stage 1: The Light-Dependent Reactions
- Location: Thylakoid membranes of the chloroplast.
- Inputs: Light, $H_2O$, $NADP^+$, ADP.
- Outputs: $O_2$, ATP, NADPH.
The Process:
- Photosystem II (PSII): Chlorophyll absorbs light energy, exciting electrons. $H2O$ is split (photolysis) to replace these electrons, releasing $O2$ as a byproduct.
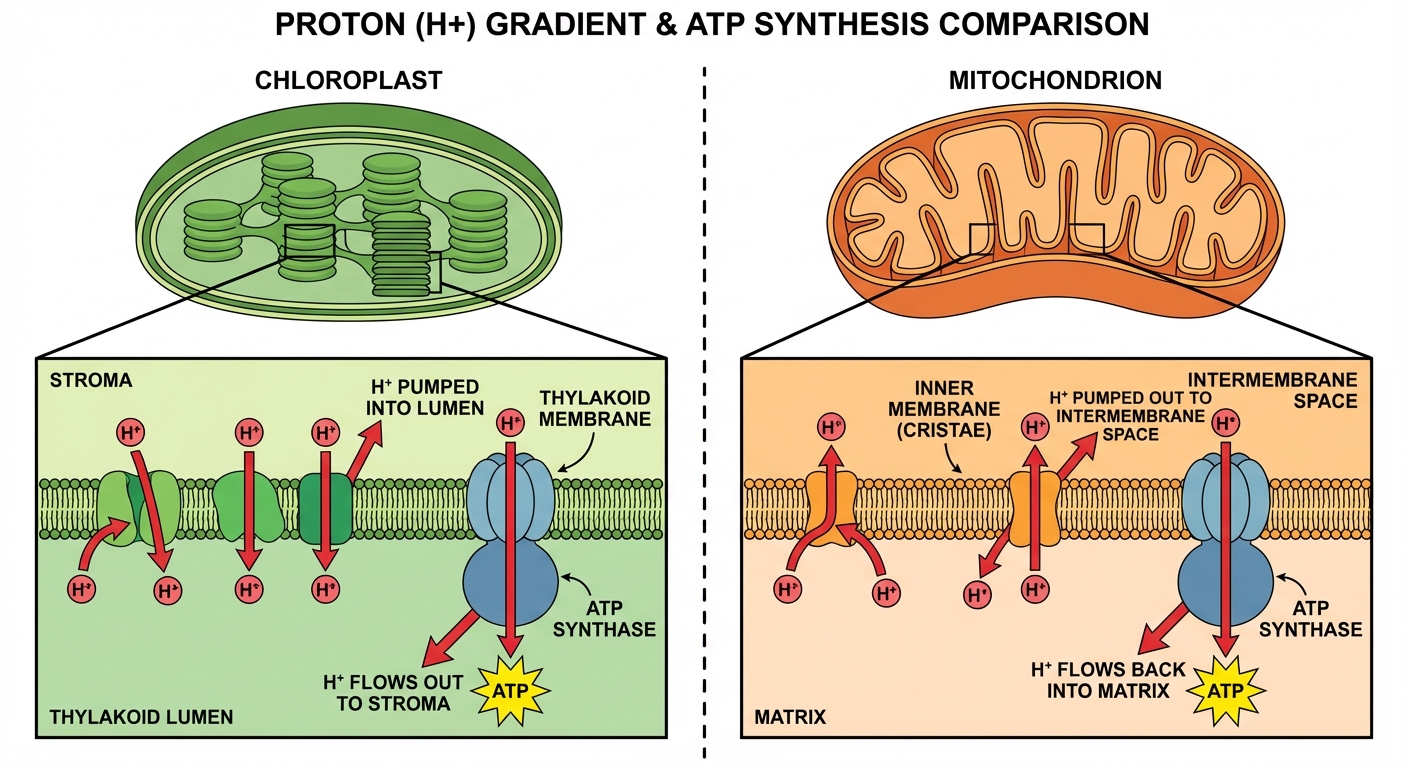
- Electron Transport Chain (ETC): Excited electrons travel down the ETC. Their energy is used to pump protons ($H^+$) from the stroma into the thylakoid lumen, creating a gradient.
- Photosystem I (PSI): Electrons are re-energized by light and ultimately transferred to $NADP^+$ to form NADPH (an electron carrier).
- ATP Synthesis: Protons flow back into the stroma through ATP Synthase (chemiosmosis), generating ATP.
Stage 2: The Calvin Cycle (Light-Independent)
- Location: Stroma of the chloroplast.
- Inputs: $CO_2$, ATP, NADPH.
- Outputs: G3P (sugar precursor), ADP, $NADP^+$.
The Process:
- Carbon Fixation: The enzyme Rubisco attaches $CO_2$ to RuBP (a 5-carbon sugar).
- Reduction: ATP and NADPH reduce the molecules to form G3P (Glyceraldehyde-3-phosphate). Two G3P molecules are required to make one glucose.
- Regeneration: Remaining G3P is used to regenerate RuBP so the cycle can continue.
Note on Adaptations: Use of $C4$ and CAM pathways allows plants in hot/arid climates to minimize water loss and photorespiration (wasteful fixation of $O2$ instead of $CO2$) by separating fixation either spatially ($C4$) or temporally (CAM).
Cellular Respiration
Definition: The release of energy from the breakdown of organic molecules (glucose) to synthesize ATP.
General Formula:.
Step 1: Glycolysis
- Location: Cytoplasm (Cytosol).
- Requirement: Occurs with or without Oxygen.
- Events: Breaks 1 Glucose (6C) into 2 Pyruvate (3C).
- Yield: Net gain of 2 ATP and 2 NADH.
Step 2: Pyruvate Oxidation (Link Reaction)
- Location: Moving into the Mitochondrial Matrix.
- Events: Pyruvate is converted to Acetyl-CoA (2C). $CO_2$ is released.
Step 3: The Krebs Cycle (Citric Acid Cycle)
- Location: Mitochondrial Matrix.
- Events: Acetyl-CoA is dismantled. Byproducts are released.
- Yield (per glucose): 2 ATP, 6 NADH, 2 $FADH2$, and $4 CO2$ (waste).
- Key Concept: The main purpose is to load electron carriers (NADH and $FADH_2$) for the next step.
Step 4: Oxidative Phosphorylation
This step produces the vast majority of ATP (~30-34 ATP).
Electron Transport Chain (ETC):
- NADH and $FADH_2$ drop off high-energy electrons at proteins in the inner mitochondrial membrane (cristae).
- Electrons move down the chain toward Oxygen, the final electron acceptor. Oxygen combines with electrons and $H^+$ to form Water.
- Crucial difference: If Oxygen is not present, the chain acts like a traffic jam; electrons back up, and the process stops.
Chemiosmosis:
- Energy from the ETC pumps $H^+$ ions from the matrix into the intermembrane space.
- This establishes the proton motive force.
- Protons flow back into the matrix through ATP Synthase, powering the bonding of ADP + $P_i \to$ ATP.
Anaerobic Respiration and Fermentation
What happens when there is no oxygen? The ETC stops, and the Krebs cycle halts. However, Glycolysis can continue if $NAD^+$ is regenerated.
Fermentation:
An extension of glycolysis that allows cells to generate ATP without oxygen. The primary purpose is to regenerate $NAD^+$ so glycolysis does not shut down.
- Alcoholic Fermentation: Pyruvate $\to$ Ethanol + $CO_2$ (Yeast, bacteria).
- Lactic Acid Fermentation: Pyruvate $\to$ Lactate (Muscle cells, some bacteria).
Fitness and Variation
Variation in molecular structure impacts survival.
- Endothermy vs. Ectothermy: Some organisms can "uncouple" oxidative phosphorylation from ATP synthesis (e.g., Brown Fat). The protons leak back across the membrane without generating ATP, generating heat instead. This is vital for hibernation and body temperature regulation.
Memory Aids & Mnemonics
- LEO the lion says GER:
- Lose Electrons = Oxidation (NADH $\to$ $NAD^+$)
- Gain Electrons = Reduction ($NAD^+$ $\to$ NADH)
- Photosynthesis vs Respiration Locations:
- Photo = Thylakoid (Tiny green pancakes)
- Respiration = Matrix (Middle of mitochondria)
Common Mistakes & Pitfalls
- Confusing Plants and Respiration: Students often think plants only do photosynthesis. WRONG. Plants have mitochondria and perform cellular respiration day and night to power their cells. They only photosynthesize when light is capable.
- Proton Gradients: Confusing the direction of pumping.
- Mitochondria: Pump OUT to Intermembrane space.
- Chloroplast: Pump IN to Thylakoid lumen.
- Tip: Always think of the gradient as "pumping into a small space" to build pressure.
- "Dark Reactions": The Calvin Cycle is often called the "Dark Reaction," leading students to think it only happens at night. It actually happens during the day because it needs the ATP/NADPH made by the light reactions.
- Energy Creation: Never say energy is "created" in the mitochondria. Say energy is transformed or ATP is synthesized.
- Role of Oxygen: Oxygen does not "burn" glucose directly. It sits at the very end of the line, waiting to catch electrons. Without it, the flow stops.