Unit 4 Complete Guide: Reaction Types, Stoichiometry, and Analysis
Introduction to Chemical Reactions
4.1 Chemical vs. Physical Changes
Definitions & Concepts
Chemical Reactions involve the breaking and forming of intramolecular bonds (ionic, covalent, metallic) to create new substances. Physical Changes involve changes in the intermolecular forces (IMFs) between molecules without changing the chemical composition of the substance.
- Physical Change: Phase changes (melting, boiling), dissolving salt in water, grinding a solid.
- Chemical Change: Rusting iron, combustion, precipitation, acid-base neutralization.
Evidence of a Chemical Reaction
While not definitive proof, the following observations suggest a chemical change has occurred:
- Production of heat or light.
- Formation of a gas (bubbles/fizzing).
- Formation of a solid precipitate.
- Distinct color change.
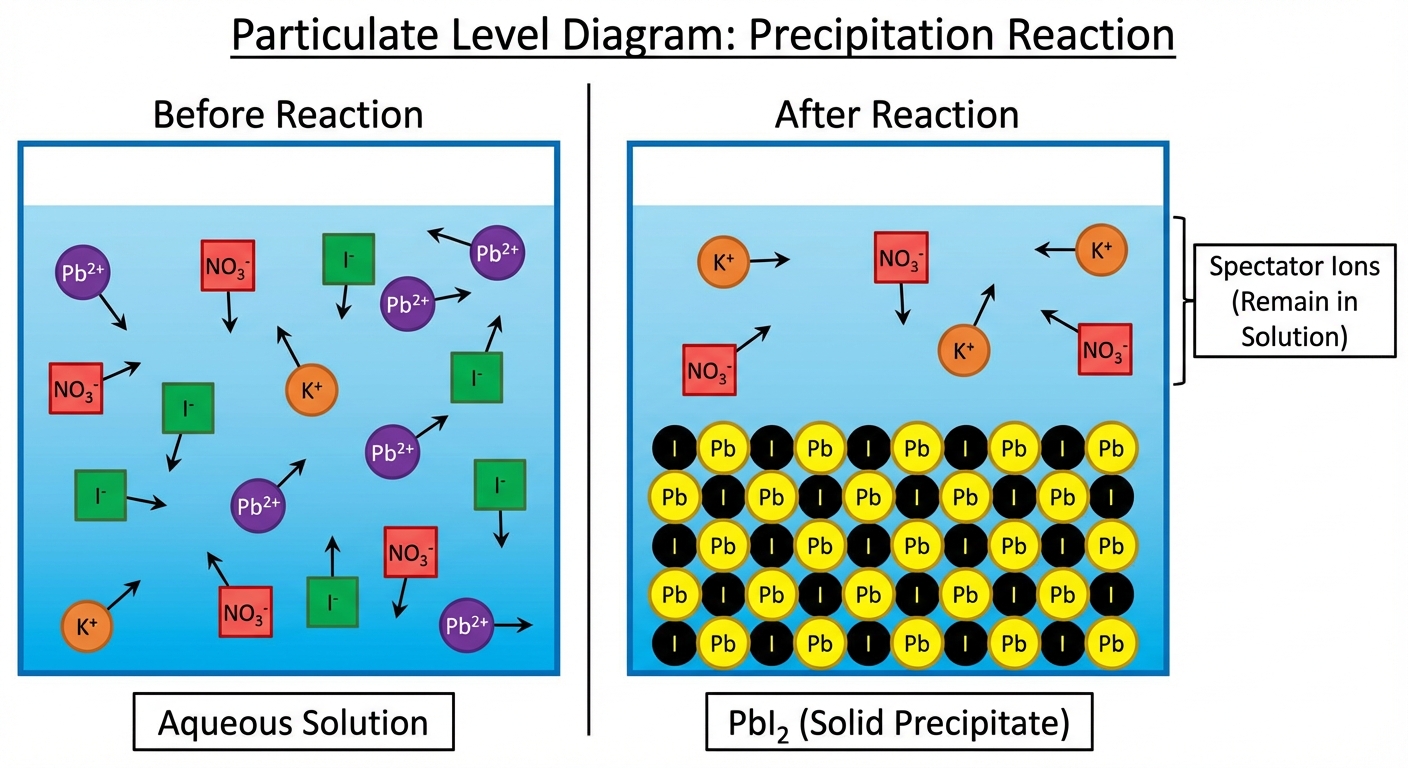
4.2 Net Ionic Equations & Solubility
The Three Levels of Equations
To truly understand reactions in aqueous solutions, we break them down into three types:
- Molecular Equation: Shows the complete formulas of all reactants and products. Useful for stoichiometry.
- Complete Ionic Equation: Shows all strong electrolytes (soluble salts, strong acids, strong bases) dissociated into ions. Solids, liquids, and gases remain intact.
- Net Ionic Equation: Removes Spectator Ions (ions that appear unchanged on both sides) to show only the species actually reacting.
Solubility Rules (The "SNAP" Rule)
For AP Chemistry, you do not need to memorize every solubility rule, but you MUST memorize that salts containing the following ions are ALWAYS soluble (no exceptions):
- S - Sodium ()
- N - Nitrate ()
- A - Ammonium ()
- P - Potassium ()
Common Mistakes
- Splitting Weak Acids: Weak acids (like or ) are weak electrolytes. In ionic equations, they must be written as whole molecules, not separated ions.
- Forgetting Charges: Omission of ionic charges in net ionic equations is a frequent point deduction.
4.3 Representations of Reactions
At the particulate level, reactions must conserve mass and atoms. AP questions frequently ask you to draw molecules in a box before and after a reaction.
Key Principles:
- Conservation of Atoms: The number of atoms of each element must remain constant.
- Orientation: For a reaction to occur, molecules must collide with the correct orientation and sufficient energy (Collision Theory).
4.5 Stoichiometry
Balancing Chemical Equations
Stoichiometry is the mathematics of chemical reactions based on the Law of Conservation of Mass.
Steps to Solve:
- Balance the Equation: Ensure atom counts match on both sides.
- Convert to Moles: Always convert given mass, volume, or concentration to moles.
- Mole Ratios: Use the coefficients from the balanced equation.
- Convert to Answer: Convert moles back to the requested unit (grams, liters, etc.).
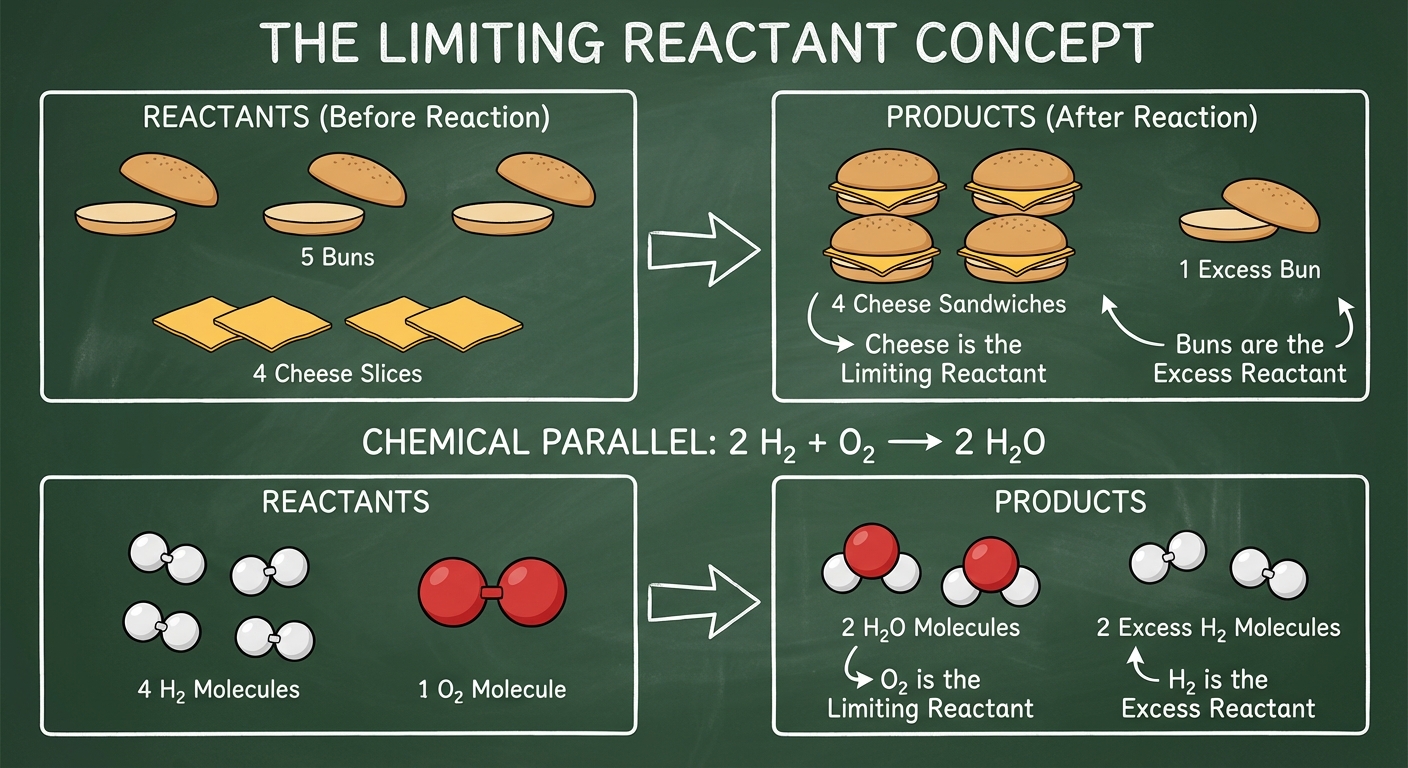
Limiting Reactants & Percent Yield
The Limiting Reactant is the reagent that is completely consumed first, limiting the amount of product formed. The Excess Reactant is left over.
How to determine the Limiting Reactant:
- Calculate the moles of each reactant.
- Divide the moles of each reactant by its coefficient in the balanced equation.
- The smallest resulting number corresponds to the limiting reactant.
Percent Yield Formula:
Combustion Analysis
Hydrocarbon combustion generally follows: .
- Carbon Rule: All Carbon in the original compound ends up in the .
- Hydrogen Rule: All Hydrogen in the original compound ends up in the .
- Oxygen Logic: Oxygen in the product comes from both the original compound and the added , so find oxygen by subtraction after finding C and H.
4.7 Types of Chemical Reactions
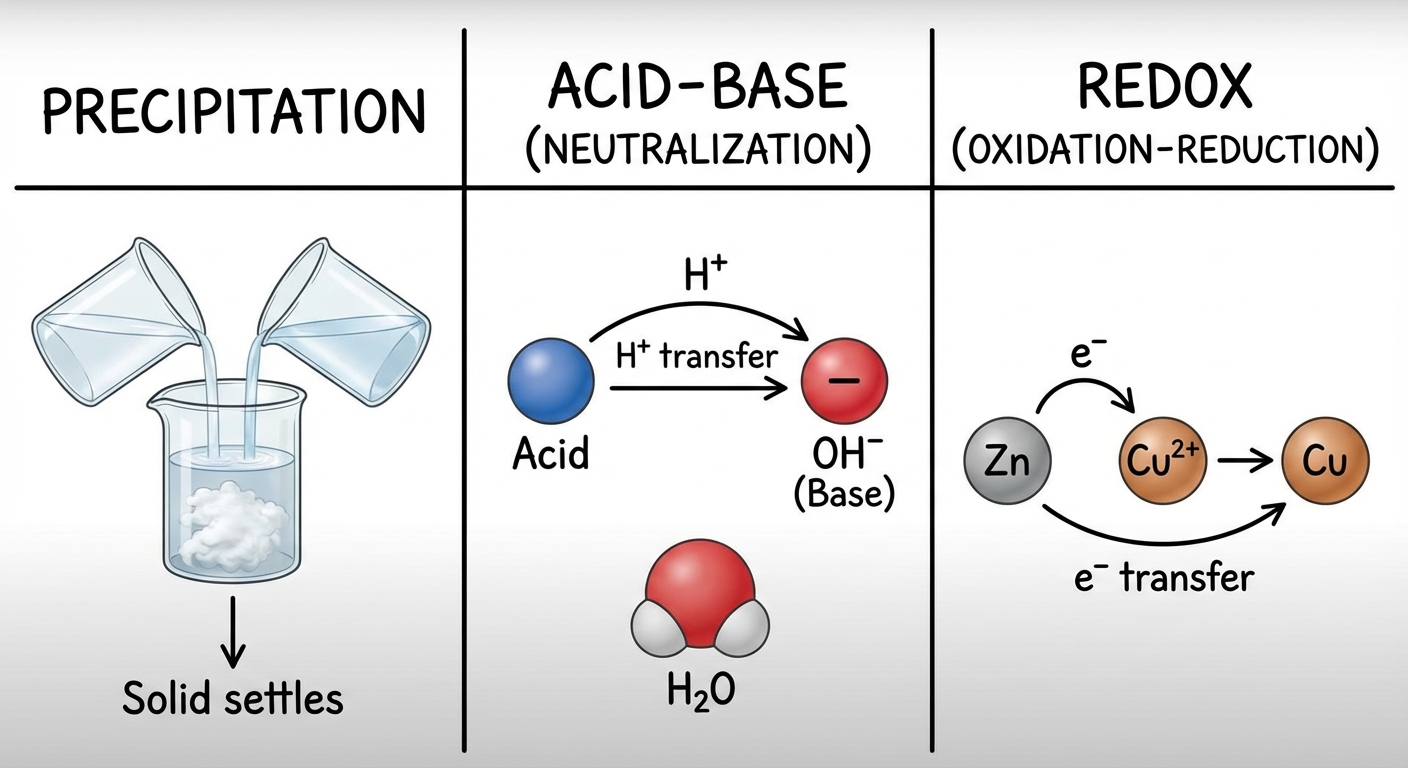
1. Precipitation Relations
Occurs when two aqueous solutions mix to form an insoluble solid (precipitate). This is a double replacement reaction.
- General form:
2. Acid-Base Reactions (Neutralization)
Definitons (Bronsted-Lowry):
- Acid: A proton () donor.
- Base: A proton () acceptor.
Conjugate Acid-Base Pairs:
Reactions involve the transfer of a proton. What remains of the acid after donating is the conjugate base.
Example:
- Acid: (Donates )
- Base: (Accepts )
- Conjugate Base:
- Conjugate Acid:
Amphoteric: Substances like water that can act as either an acid or a base depending on the environment.
3. Oxidation-Reduction (Redox) Reactions
Redox reactions involve the transfer of electrons. Oxidation numbers are used to track this transfer.
Mnemonic: LEO the lion says GER
- Lose Electrons = Oxidation (Oxidation number increases)
- Gain Electrons = Reduction (Oxidation number decreases)
Rules for Oxidation States:
- Elements: Neutral atoms (e.g., , ) are 0.
- Monatomic Ions: Equal to their charge (e.g., is +1).
- Oxygen: Usually -2 (Exceptions: Peroxides like are -1).
- Hydrogen: +1 with nonmetals, -1 with metals.
- Fluorine: Always -1.
Example:
- : Goes from 0 to +3 (Oxidized)
- : Goes from +1 to 0 (Reduced)
- : Remains -1 (Spectator)
4.6 & 4.8 Chemical Analysis Techniques
Titrations
A laboratory method to determine the concentration of an unknown analyte using a known titrant.
- Equivalence Point: The point where the moles of titrant stoichiometrically equal the moles of analyte (perfect reaction).
- End Point: The point where the indicator changes color (should be close to equivalence point).
Calculations:
For a 1:1 mole ratio (e.g., ):
For ratios other than 1:1, use dimensional analysis (stoichiometry).
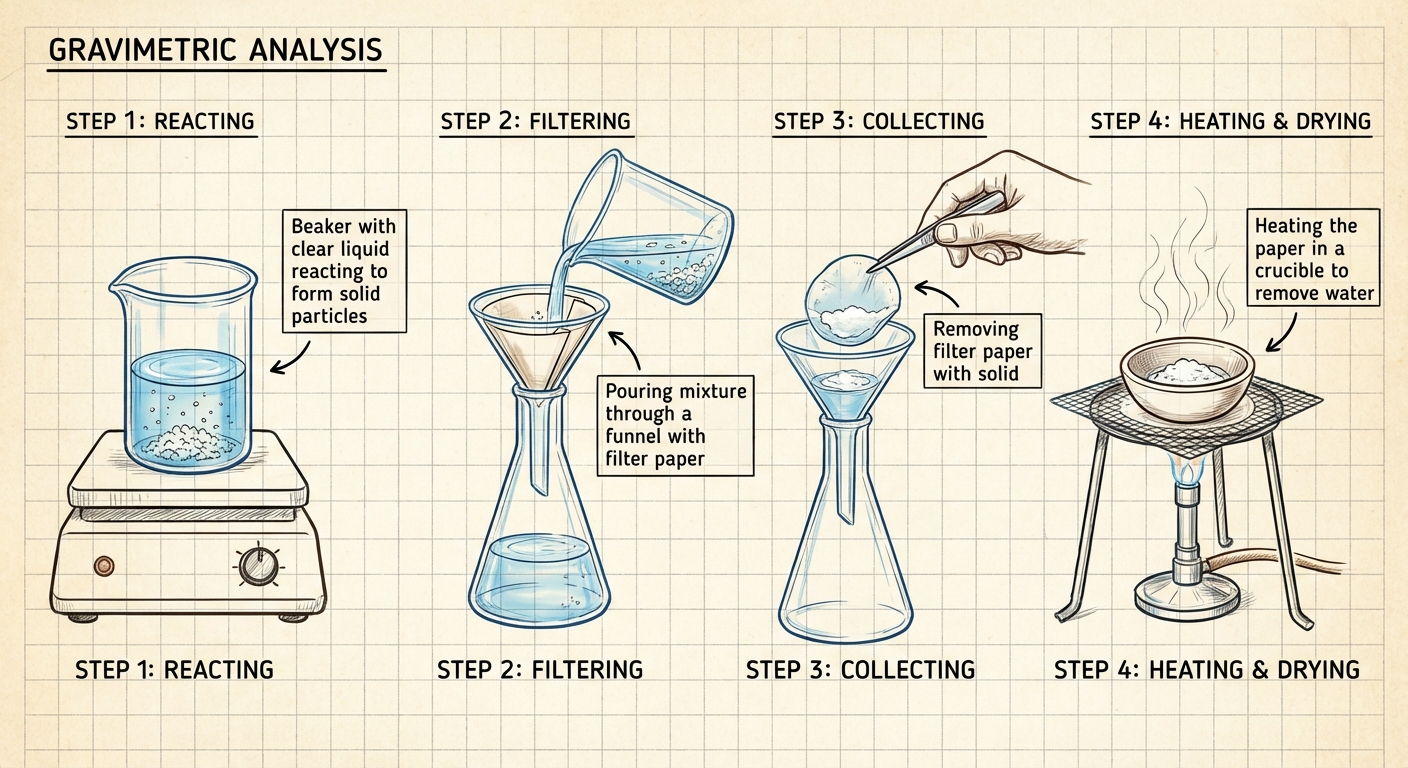
Gravimetric Analysis
Using mass measurements to determine the amount of an analyte. The analyte is usually precipitated out of solution, filtered, dried, and weighed.
WORKED PROBLEM: Identifying an Unknown
Problem: A 4.33 g sample of an unknown alkali hydroxide () is dissolved. Excess Copper(II) Nitrate is added to precipitate Copper(II) Hydroxide according to:
The dried precipitate has a mass of 3.81 g. Identify the alkali metal ().
Solution:
Convert Precipitate Mass to Moles:
- Molar Mass of
Use Stoichiometry to find moles of Unknown:
- Ratio is 2:1 ()
Determine Molar Mass of Unknown:
Identify Element M:
- Mass of
- Looking at the periodic table, Potassium () is 39.10 g/mol. Sodium () is 22.99 g/mol.
- Answer: The unknown is likely Potassium (KOH).
Common Mistakes in Analysis
- Titration: Misreading the buret (volume is read top-down). Forgetting to rinse the buret with the titrant before starting (which dilutes the titrant, causing over-titration).
- Gravimetric: Not drying the precipitate completely. If water remains, the mass is too high, leading to a calculation indicating more moles of product than actually exist.
Summary of Common Pitfalls
- Redox: Confusing oxidation state with ionic charge. (Oxidation state is a formalism; charge is a physical property).
- Solubility: Assuming common ions like are always soluble (, , are insoluble).
- Stoichiometry: Forgetting to balance the equation before doing the math.
- Gases: Forgetting that , , , , , , and are diatomic when found as elements in a reaction.