Unit 2 Guide: Molecular and Ionic Compound Structure and Properties
Mastering Molecular Architecture: Lewis Structures & VSEPR
To understand how matter interacts, we must visualize how atoms connect. This section covers the blueprint of chemistry: drawing molecules (Lewis Structures), evaluating their stability (Formal Charge), and predicting their 3D shape (VSEPR).
constructing Lewis Diagrams
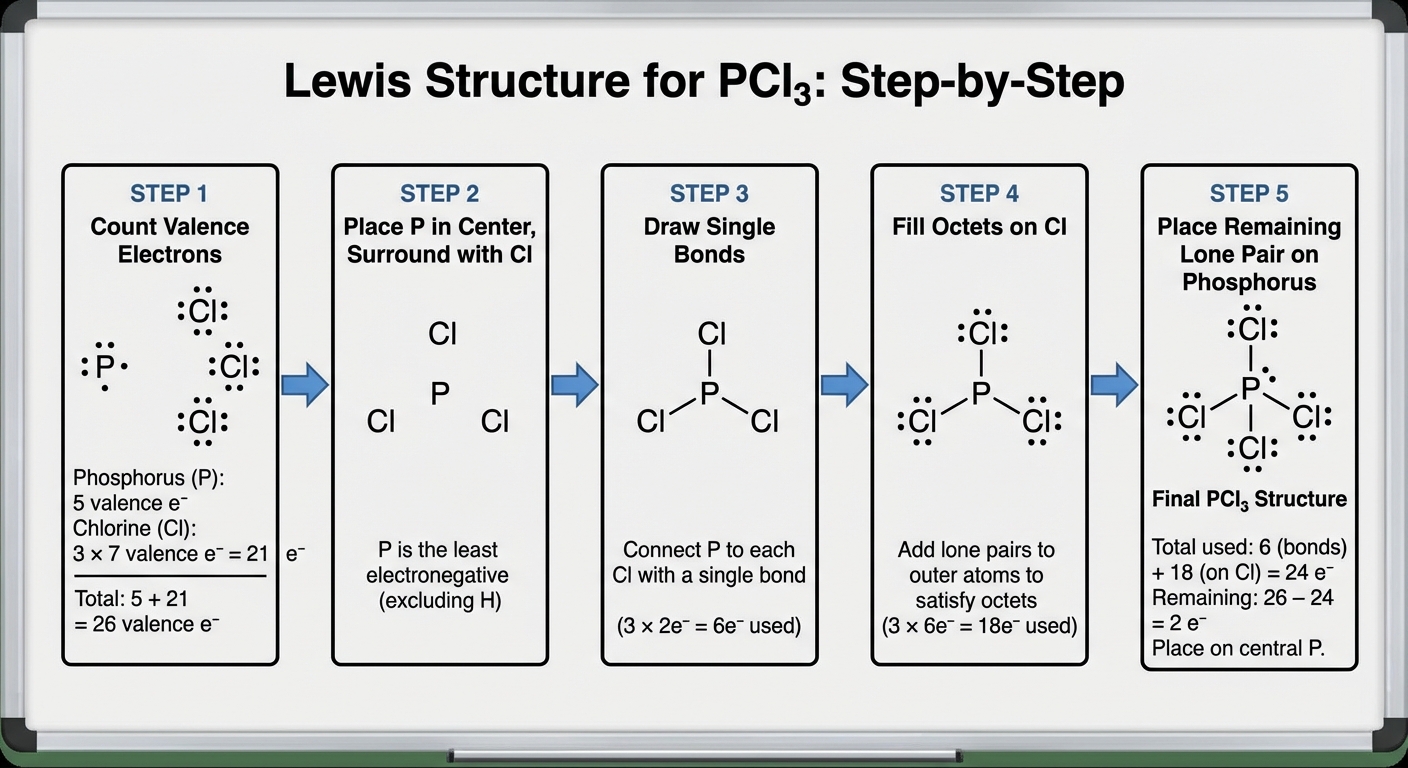
A Lewis Diagram is a two-dimensional representation of a molecule showing how valence electrons are distributed among atoms as bonds or lone pairs.
Generel Rules for Drawing
Follow these steps to construct a valid structure:
- Count Valence Electrons: Sum the valence electrons for all atoms.
- For Polyatomic Ions: Add electrons for negative charges; subtract for positive charges.
- Determine the Central Atom: Usually the least electronegative element (Equation: never Hydrogen). Carbon is almost always central.
- Draw Single Bonds: Connect the central atom to surrounding atoms (deduct 2 electrons per bond from your total).
- Complete Octets (Surrounding Atoms): Add lone pairs to outer atoms first to satisfy the Octet Rule (8 valence electrons).
- Finish Central Atom: Place remaining electrons on the central atom, even if it exceeds the octet (see exceptions below).
- Check Octets & Form Multiple Bonds: If the central atom lacks an octet, convert lone pairs from outer atoms into double or triple bonds.
Important Exceptions to the Octet Rule
Not every atom follows the "Rule of 8."
- Incomplete Octets:
- Hydrogen (H): Stable with 2 electrons (duet rule).
- Boron (B): often stable with 6 electrons (e.g., $BF_3$).
- Beryllium (Be): often stable with 4 electrons (e.g., $BeCl_2$).
- Expanded Octets (Hypervalency):
- Elements in Period 3 and below (P, S, Cl, Xe, etc.) have access to empty d-orbitals, allowing them to hold more than 8 electrons (e.g., $PCl5$, $SF6$).
Resonance and Formal Charge
Sometimes, a single Lewis structure is insufficient to describe a molecule.
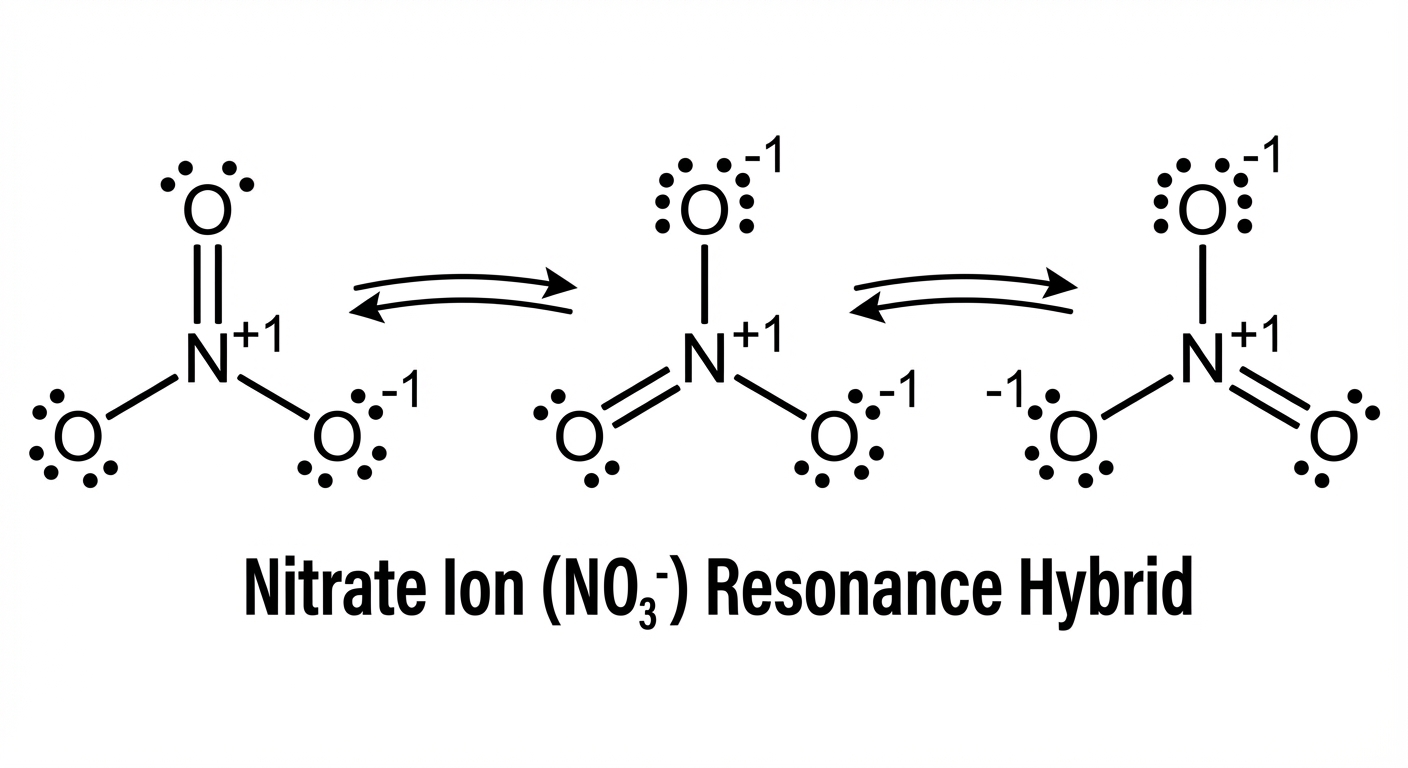
Resonance Structures
Resonance occurs when equivalent Lewis structures can be drawn for the same molecule.
- Concept: The electrons are not flipping back and forth between positions; they are delocalized (spread out).
- Bond Order: The actual bond is a hybrid. For example, in Ozone ($O_3$), the bond is a blend of a single and double bond (bond order 1.5).
Formal Charge
Formal Charge (FC) is a bookkeeping method used to determine the dominant (most stable) Lewis structure when multiple valid options exist.
Where:
- $V$ = Number of Valence electrons (from periodic table)
- $N$ = Number of Nonbonding electrons (individual dots)
- $B$ = Number of Bonding electrons (lines $\times$ 2)
Simplified Calculation:
Rules for Selecting the Best Structure
- The structure where formal charges are closest to zero is preferred.
- The sum of formal charges must equal the overall charge of the molecule or ion.
- If a negative formal charge is necessary, it must reside on the most electronegative atom.
Example: $CO_2$
- Structure A (O=C=O): O has 6 valence, 4 dots, 2 bonds. $6 - 4 - 2 = 0$. C has $4 - 0 - 4 = 0$. All zeros. (Stable)
- Structure B (O-C≡O): Single bond O is $-1$, Triple bond O is $+1$. Less stable due to charge separation.
VSEPR and Bond Hybridization
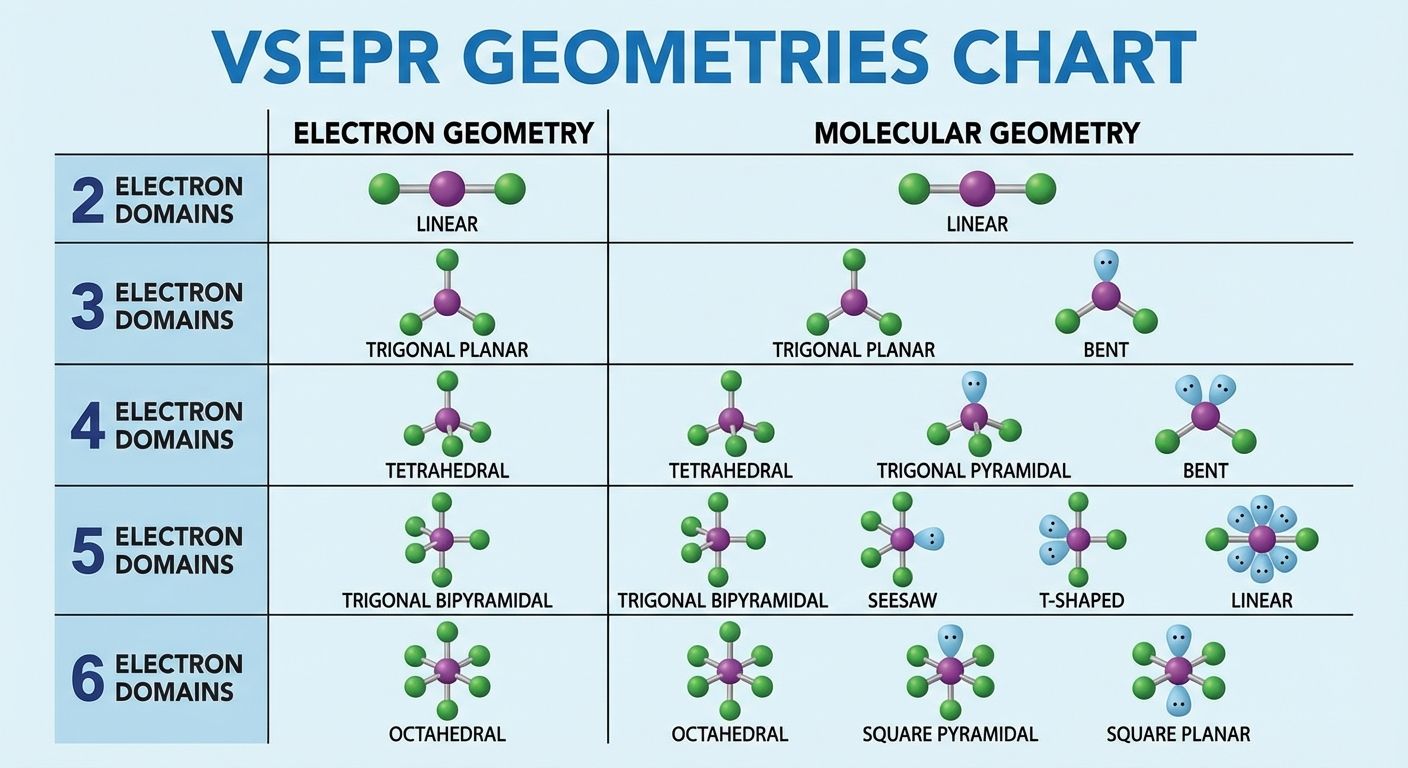
Once the 2D Lewis structure is defined, we use VSEPR (Valence Shell Electron Pair Repulsion) theory to predict the 3D geometry. The core principle: Electron domains (bonds and lone pairs) repel each other and try to get as far apart as possible.
Electron Domains & Repulsion Order
An "Electron Domain" is a region of electron density.
- Note: A single, double, or triple bond counts as one domain.
Repulsion Hierarchy:
Lone:Pair-Lone:Pair > Lone:Pair-Bonding:Pair > Bonding:Pair-Bonding:Pair
Because lone pairs occupy more space, they compress bond angles slightly (e.g., Tetrahedral is $109.5^\circ$, but Ammonia ($NH_3$) with one lone pair is $\approx 107^\circ$).
Geometry Summary Table
| Domains | Electron Geometry | Bonding / Lone Pairs | Molecular Geometry | Approx Angle | Hybridization |
|---|---|---|---|---|---|
| 2 | Linear | 2 / 0 | Linear | $180^\circ$ | $sp$ |
| 3 | Trigonal Planar | 3 / 0 | Trigonal Planar | $120^\circ$ | $sp^2$ |
| 2 / 1 | Bent | $<120^\circ$ | |||
| 4 | Tetrahedral | 4 / 0 | Tetrahedral | $109.5^\circ$ | $sp^3$ |
| 3 / 1 | Trigonal Pyramidal | $<109.5^\circ$ | |||
| 2 / 2 | Bent | $<109.5^\circ$ | |||
| 5 | Trigonal Bipyramidal | 5 / 0 | Trigonal Bipyramidal | $90^\circ, 120^\circ$ | $dsp^3$ |
| 4 / 1 | Seesaw | $<90^\circ, <120^\circ$ | |||
| 3 / 2 | T-Shaped | $<90^\circ$ | |||
| 2 / 3 | Linear | $180^\circ$ | |||
| 6 | Octahedral | 6 / 0 | Octahedral | $90^\circ$ | $d^2sp^3$ |
| 5 / 1 | Square Pyramidal | $<90^\circ$ | |||
| 4 / 2 | Square Planar | $90^\circ$ |
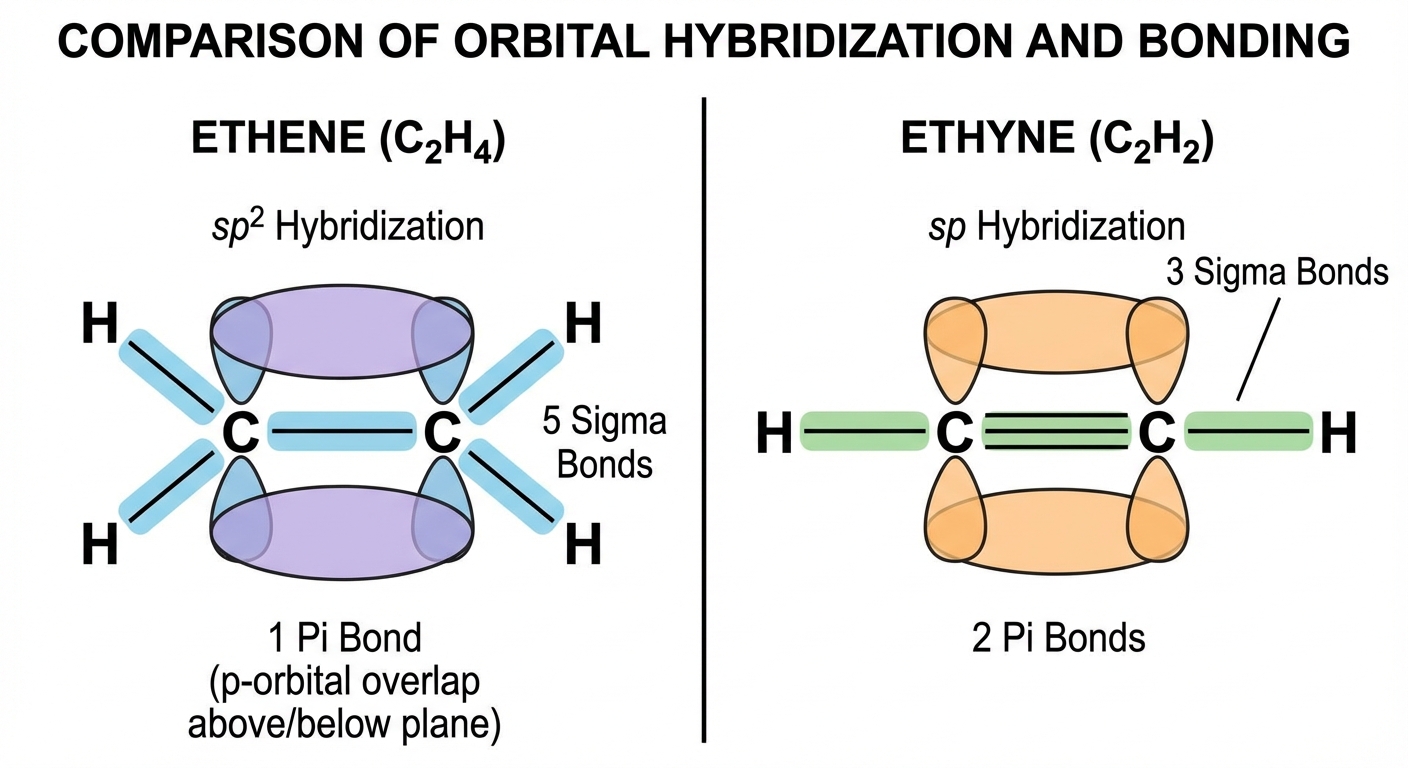
Bond Hybridization
Hybridization explains how atomic orbitals mix to form equal energy bonding orbitals.
The Trick: Count the electron domains (steric number) around the central atom.
- 2 Domains $\rightarrow$ $sp$ (one s + one p)
- 3 Domains $\rightarrow$ $sp^2$ (one s + two p)
- 4 Domains $\rightarrow$ $sp^3$ (one s + three p)
Sigma ($\sigma$) and Pi ($\pi$) Bonds
- Sigma ($\sigma$) Bond: Direct head-to-head overlap. Every single bond is a sigma bond. Free rotation is possible.
- Pi ($\pi$) Bond: Sideways overlap of unhybridized p-orbitals. Weaker than sigma. NO free rotation.
Counting Rules:
- Single Bond = $1\sigma$
- Double Bond = $1\sigma + 1\pi$
- Triple Bond = $1\sigma + 2\pi$
Common Mistakes & Pitfalls
- Confusing Molecular vs. Electron Geometry: The Electron geometry includes lone pairs in the name (e.g., Tetrahedral). The Molecular geometry ignores them visually but accounts for their repulsion (e.g., Bent). Always read the question carefully to see which one it asks for.
- Forgetting Lone Pairs on Outer Atoms: Even though they don't affect central geometry, forgetting to draw dots on Chlorine or Fluorine results in an incorrect electron count.
- Applying Resonance Incorrectly: Students often move atoms to create resonance. Only electrons (pi bonds and lone pairs) move; atoms stay fixed.
- Miscalculating Formal Charge: Remember, bonds count as 1 electron for the atom, and lone pairs count as 2. Don't subtract the total electrons in the bond.
- Assuming Water is $109.5^\circ$: Because water has two lone pairs, the compression is significant. The angle is approximately $104.5^\circ$, not the perfect tetrahedral $109.5^\circ$.