Comprehensive Guide to Chemical Bonding and Molecular Geometry
Unit Overview: Structure and Bonding
Chemical bonds are the attractive forces that hold atoms together. In this unit, we explore intramolecular forces—the forces affecting the structure within a single molecule or ionic lattice. The type of bond formed depends largely on the electronegativity differences between atoms and the desire to achieve a stable, lowest-energy electron configuration (usually a noble gas configuration).
2.1 Types of Chemical Bonds
Chemical bonds exist on a continuum based on the difference in electronegativity ($\Delta\chi$) between the bonding atoms.
Electronegativity and Bond Character
- Nonpolar Covalent: Electrons are shared equally. Usually $\Delta\chi < 0.4$. (e.g., $Cl-Cl$, $C-H$).
- Polar Covalent: Electrons are shared unequally. The more electronegative atom gains a partial negative charge ($\delta-$), and the other gains a partial positive charge ($\delta+$). Usually $0.4 < \Delta\chi < 1.7$.
- Ionic: Electrons are transferred completely from a metal to a nonmetal. usually $\Delta\chi > 1.7$.
The Bond Spectrum
It is important to remember that bonding is rarely 100% ionic or 100% covalent; it is a spectrum of percent ionic character. As the electronegativity difference increases, the polarity of the bond increases.
2.2 Intramolecular Potential Energy
Covalent bonds form at specific distances where the system achieves the lowest possible potential energy. This is a balance between:
- Attraction: Nucleus of atom A attracting electrons of atom B.
- Repulsion: Nucleus A repelling Nucleus B, and electrons of A repelling electrons of B.
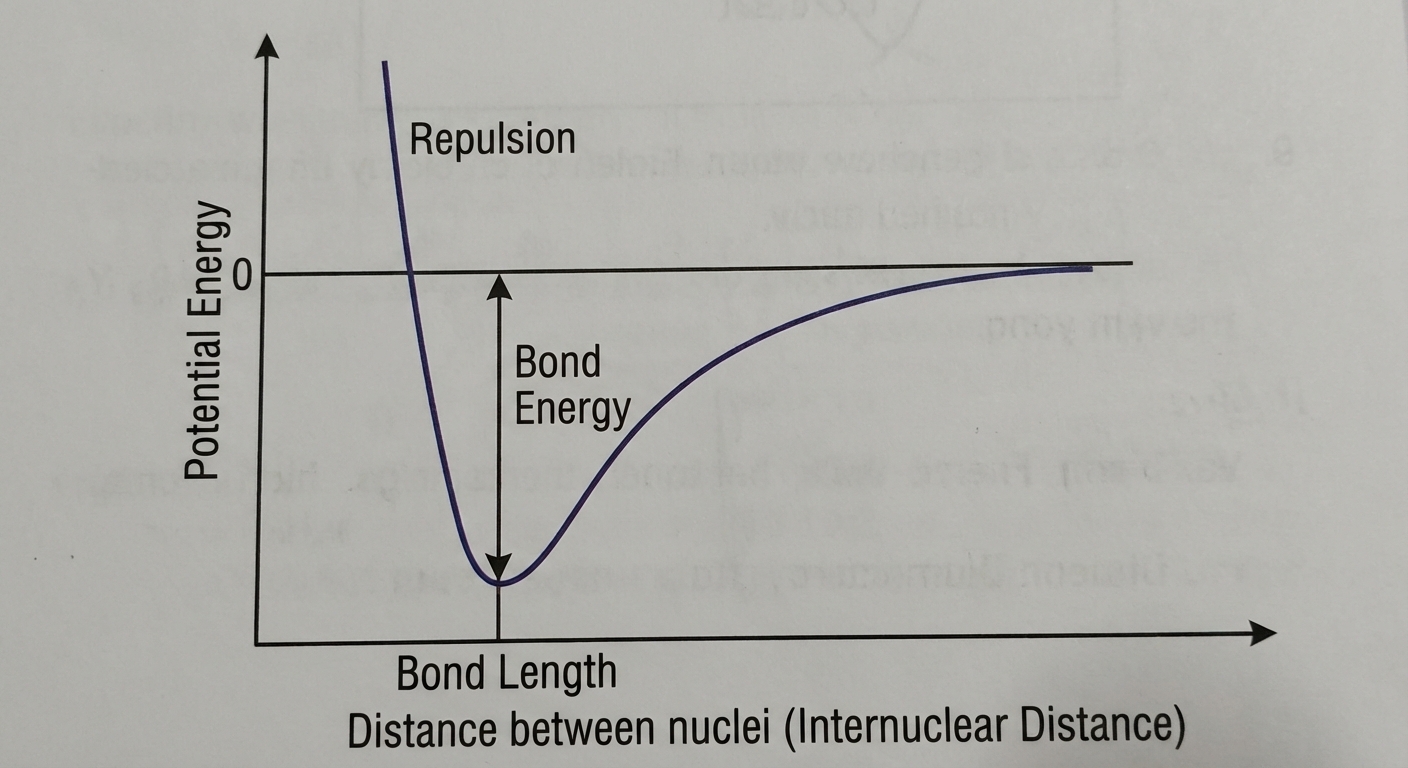
Interpreting the Graph
- Infinite Distance ($PE = 0$): Atoms are too far apart to interact.
- The Well (Minimum $PE$): This is the Bond Length. The attractive and repulsive forces are balanced.
- Steep Rise (Positive $PE$): Atoms are too close; internuclear repulsion dominates.
2.3 Ionic Compounds and Solids
Ionic bonds result from the electrostatic attraction between cations (positive) and anions (negative) organized in a rigid crystalline lattice.
Properties of Ionic Solids
- structure: Crystal Lattice (alternating $+$ and $-$ ions).
- State: Solid at room temperature.
- Melting/Boiling Point: Very high (requires breaking strong electrostatic forces).
- Volatility: Very low (do not vaporize easily).
- Brittleness: Hard but brittle. If struck, ions of like charge align and repel, shattering the crystal.
Conductivity Rules
The mobility of charged particles (ions) determines conductivity.
- Solid: No. Ions are locked in the lattice.
- Liquid (Molten): Yes. Lattice is broken; ions can slide past each other.
- Aqueous (Dissolved): Yes. Water separates ions, allowing free movement.
Lattice Energy and Coulomb’s Law
Lattice Energy is the energy released when ions form a stable crystal lattice. The strength of an ionic bond is governed by Coulomb’s Law:
Where $Q$ is the charge of ions and $r$ is the distance between nuclei (ionic radii).
- Charge Magnitude ($Q$): This is the most significant factor. Higher charges = Stronger attraction = Higher Melting Point.
- Example: $MgO$ ($+2/-2$) has a much higher melting point than $NaCl$ ($+1/-1$).
- Ionic Radius ($r$): Smaller ions can get closer together, leading to stronger attraction.
- Example: $NaCl$ has a stronger bond than $KCl$ because $Na^+$ is smaller than $K^+$.
2.4 Structure of Metals and Alloys
Metallic Bonding
Metals consist of a lattice of positive cores (cations) floating in a "sea of delocalized electrons." These valence electrons are not fixed to any specific atom.
- Conductivity: Excellent (solids and liquids) because electrons are free to move.
- Malleability/Ductility: Atoms can slide past one another without breaking the bond because the electron sea adjusts to the new shape.
Alloys
An alloy is a mixture of metals.
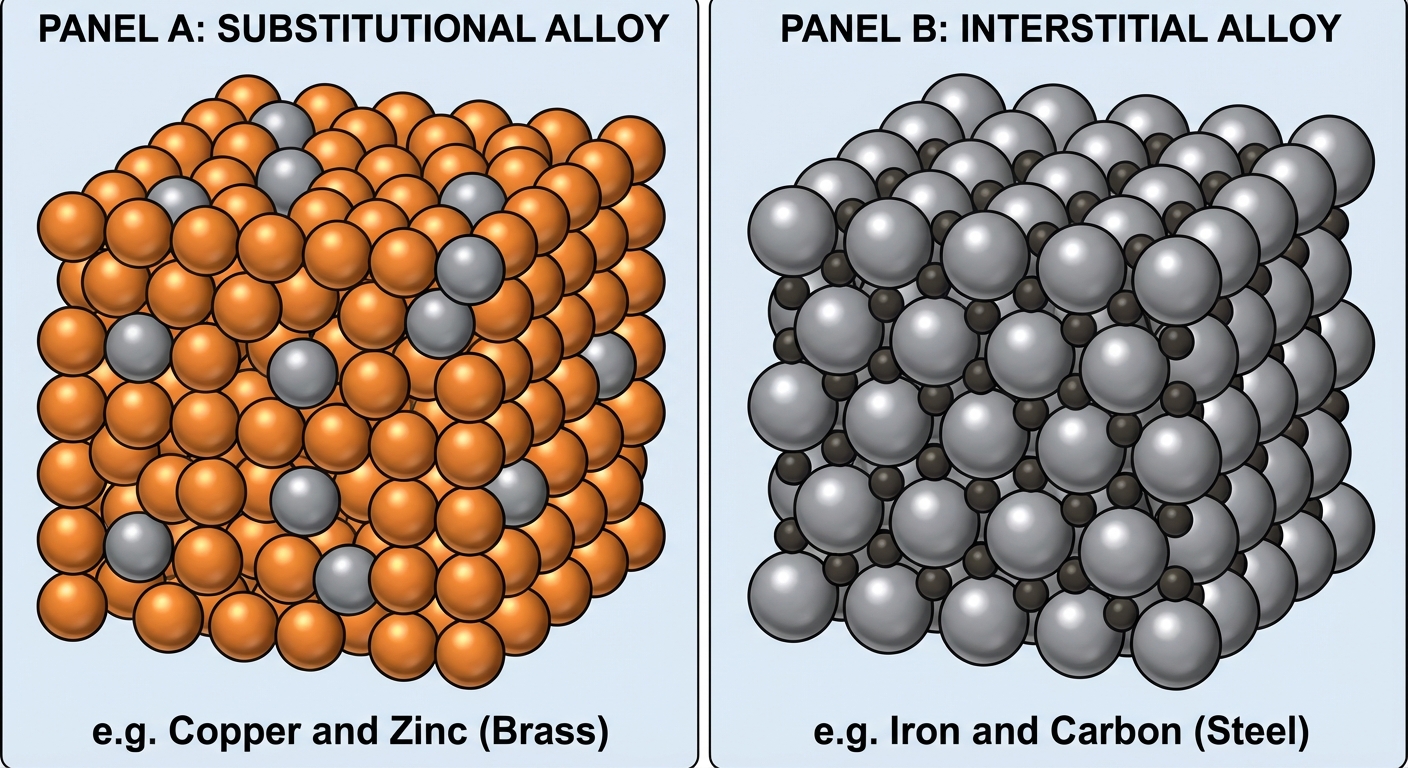
| Type | Description | Example |
|---|---|---|
| Interstitial Alloy | Small atoms fill the spaces (interstices) between larger metal atoms. Increases rigidity and makes the lattice harder to slide. | Steel (Carbon sits between Iron atoms). |
| Substitutional Alloy | Atoms of similar radius replace distinct metal atoms in the lattice. | Brass (Zinc replaces Copper; similar radii). |
2.5 & 2.6 Lewis Diagrams and Resonance
Drawing Lewis Structures
- Count Valence Electrons: Sum valence electrons for all atoms. (Add for anions, subtract for cations).
- Skeletal Structure: Place the least electronegative atom in the center (H is never central).
- Bonding: Connect atoms with single bonds (minus 2 electrons per bond from total).
- Complete Octets: Add lone pairs to outer atoms first to satisfy the Octet Rule (8 electrons).
- Dump Leftovers: Place remaining electrons on the central atom, even if it exceeds the octet (only for Period 3+ elements).
- Double/Triple Bonds: If the central atom lacks an octet, move lone pairs from outer atoms to form double/triple bonds.
Formal Charge
Formal charge helps determine the most valid structure among several possibilities. The sum of formal charges must equal the total molecule charge.
Selection Rules:
- Dominant structure has FC closest to zero for all atoms.
- If a negative FC is necessary, it should reside on the most electronegative atom.
Resonance
Resonance occurs when more than one valid Lewis structure can be drawn (often moving a double bond). The actual molecule is a hybrid (average) of all resonance structures.
- Example: In Carbonate ($CO_3^{2-}$), there is 1 double bond and 2 single bonds distributed over 3 locations.
- Bond Order: $\frac{\text{Total Bonds}}{\text{Bonding Domains}}$
- $CO_3^{2-}$ Bond Order $= 4 \text{ bonds} / 3 \text{ locations} = 1.33$.
- This implies all three bonds are identical in length and stronger than a single bond but weaker than a double bond.
Octet Exceptions
- Incomplete Octet: Boron ($B$) is stable with 6 electrons; Beryllium ($Be$) with 4.
- Expanded Octet: Elements in Period 3 or higher (e.g., $P, S, Xe$) can hold >8 electrons because of accessible d-orbitals.
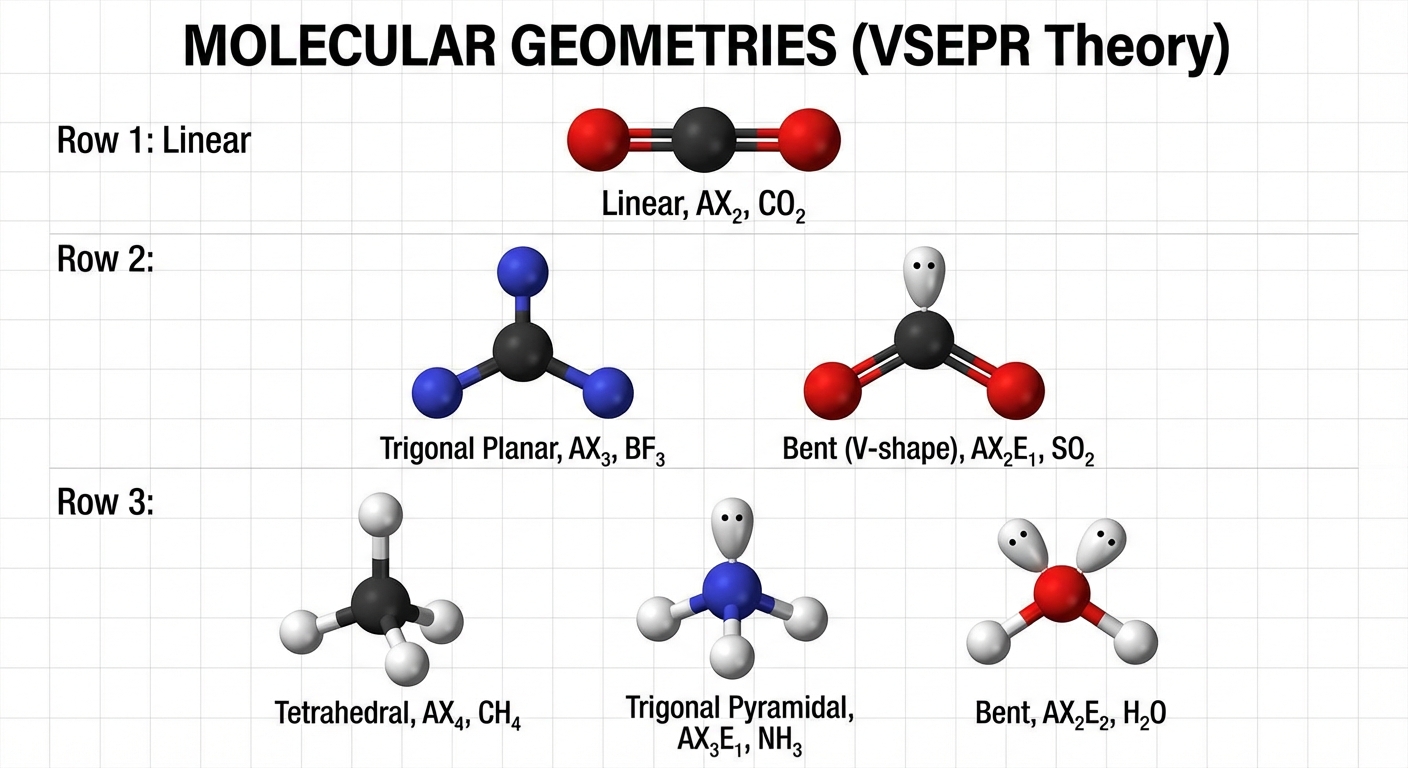
2.7 VSEPR and Molecular Geometry
Valence Shell Electron Pair Repulsion theory predicts shape based on the idea that electron domains (bonds and lone pairs) repel each other and want to be as far apart as possible.
Repulsion Hierarchy
Lone\ Pair > Triple\ Bond > Double\ Bond > Single\ Bond
Lone pairs occupy more space, compressing the bond angles slightly.
Geometry Table
| Total Domains | Bonding Pairs | Lone Pairs | Electron Geometry | Molecular Geometry | Bond Angle | Hybridization |
|---|---|---|---|---|---|---|
| 2 | 2 | 0 | Linear | Linear | $180^\circ$ | $sp$ |
| 3 | 3 | 0 | Trigonal Planar | Trigonal Planar | $120^\circ$ | $sp^2$ |
| 2 | 1 | Trigonal Planar | Bent | $<120^\circ$ | $sp^2$ | |
| 4 | 4 | 0 | Tetrahedral | Tetrahedral | $109.5^\circ$ | $sp^3$ |
| 3 | 1 | Tetrahedral | Trigonal Pyramidal | $<109.5^\circ$ | $sp^3$ | |
| 2 | 2 | Tetrahedral | Bent | $\ll 109.5^\circ$ | $sp^3$ | |
| 5 | 5 | 0 | Trig. Bipyramidal | Trig. Bipyramidal | $90^\circ, 120^\circ$ | ($dsp^3$)* |
| 4 | 1 | Trig. Bipyramidal | Seesaw | $<90^\circ, <120^\circ$ | ||
| 3 | 2 | Trig. Bipyramidal | T-Shaped | $<90^\circ$ | ||
| 2 | 3 | Trig. Bipyramidal | Linear | $180^\circ$ | ||
| 6 | 6 | 0 | Octahedral | Octahedral | $90^\circ$ | ($d^2sp^3$)* |
| 4 | 2 | Octahedral | Square Planar | $90^\circ$ |
Note: While 5 and 6 domain geometries are tested, the specific d-hybridization labels are often excluded from recent AP exams.
2.8 Bond Hybridization
Orbtials mix to form new identical hybrid orbitals to facilitate bonding.
Sigma ($\sigma$) vs Pi ($\pi$) Bonds
- Sigma ($\sigma$): Head-to-head overlap. Present in ALL covalent bonds. Free rotation allowed.
- Pi ($\pi$): Side-to-side overlap of unhybridized p-orbitals. Present in double and triple bonds. Prevents rotation (rigid).
Bond Counting:
- Single Bond: 1 $\sigma$
- Double Bond: 1 $\sigma + 1 \pi$
- Triple Bond: 1 $\sigma + 2 \pi$
- Note: Sigma bonds are stronger than Pi bonds individually, but a Double bond (Sigma + Pi) is stronger than a single bond overall.
Comparison of Properties Summary
| Property | Ionic | Network Covalent | Metallic | Polar/Nonpolar Molecular |
|---|---|---|---|---|
| Particles | Ions | Atoms | Cations in electron sea | Molecules |
| Conductivity | Liquid/Aq only | No (except graphite) | Yes (Solid/Liquid) | No |
| Melting Point | High | Very High | Variable (usually high) | Low |
| Examples | NaCl, MgO | Diamond ($C$), $SiO_2$ | Fe, Na, Cu | $H2O$, $CO2$, Sugar |
Common Mistakes & Pitfalls
- Confusing Bond Polarity with Molecular Polarity:
- A molecule can contain polar bonds but be nonpolar overall if the geometry is symmetrical and dipoles cancel out (e.g., $CF_4$ is nonpolar, despite having very polar bonds).
- Ionic "Molecules":
- Never call an ionic compound a "molecule." Refer to it as a "formula unit" or "lattice." Molecules imply covalent bonding.
- Lattice Energy Logic:
- When explaining melting points of salts, do not mention electronegativity. You MUST mention Coulomb's Law: Charge magnitude first, then ionic radius.
- Drawing Lewis Structures:
- Students often forget to count the charge of a polyatomic ion in the total valence count (e.g., forgetting the $+1$ electron for $NO_3^-$).
- Bond Angles:
- Assuming a bent molecule is always $109.5^\circ$. If there are lone pairs ($H_2O$), the angle is compressed to ~$104.5^\circ$. Always indicate logic: "Lone pair repulsion forces bonding pairs closer."